Application of Cyclin-dependent Kinase 4/6 Inhibitors in the Treatment of Malignancies and the Mechanism of Drug Resistance
-
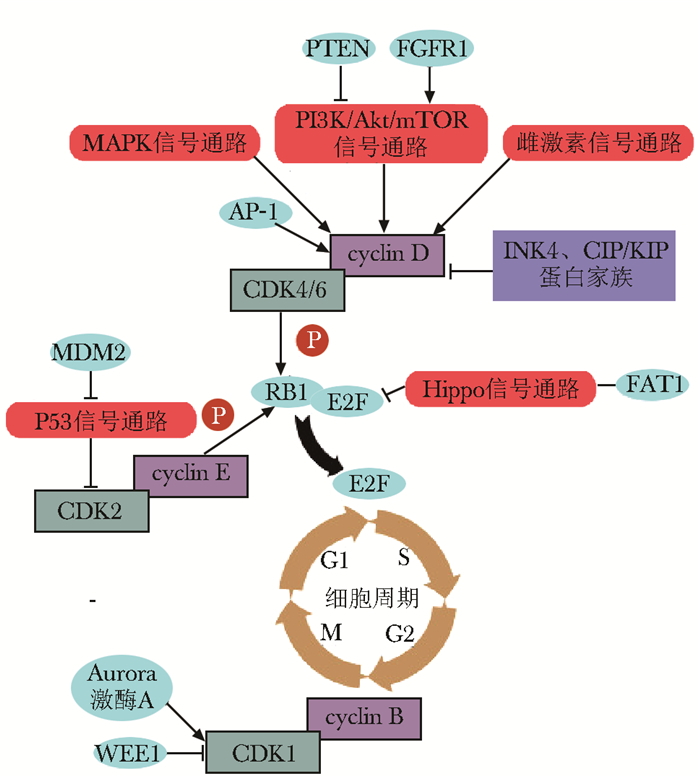
摘要: 增殖失控是恶性肿瘤的重要特征之一。细胞周期依赖性激酶4/6(cyclin-dependent kinase 4/6, CDK4/6)抑制剂能作用于各种原因导致的过度活化的CDK4/6,恢复正常细胞周期,并可通过增强免疫、改变肿瘤微环境等发挥抗肿瘤作用。目前,CDK4/6抑制剂在激素受体阳性乳腺癌治疗中取得了良好疗效,已被批准联合内分泌治疗作为此类肿瘤的一线治疗方案,在其他肿瘤中的应用亦逐渐开展,疗效有待验证。对CDK4/6抑制剂天然或获得性耐药是影响其疗效的重要因素,目前激素受体阳性(主要为雌激素受体阳性)能较为准确预测内分泌联合CDK4/6抑制剂治疗的反应性,其他标志物需进一步探索和验证。本文对CDK4/6抑制剂治疗恶性肿瘤的作用机制、应用现状及耐药机制进行梳理和总结,并对当前CDK4/6抑制剂治疗乳腺癌尚存争议的临床决策问题作简要讨论。
-
关键词:
- 细胞周期依赖性激酶4/6抑制剂 /
- 细胞周期调控 /
- 乳腺癌 /
- 治疗 /
- 耐药机制
Abstract: Uncontrolled cell proliferation is one of the important hallmarks of malignancies. Cyclin-dependent kinase 4/6 (CDK4/6) inhibitors act on CDK4/6 that is over activated by various causes. CDK4/6 inhibitors play anti-tumor roles by restoring normal cell cycle, enhancing anti-tumor immunity, and reforming tumor microenvironment. Currently, CDK4/6 inhibitors have been confirmed to be effective in hormone-receptor-positive breast cancer and were approved as the first-line treatment for this breast cancer in combination with endocrine therapy. Furthermore, CDK4/6 inhibitors have been gradually applied to the treatment of other tumors, but the efficacy remains to be evaluated. Natural or acquired resistance to CDK4/6 inhibitors is a major factor affecting their efficacy. However, at present, only hormone receptor-positive (mainly estrogen receptor-positive) can relatively accurately predict the responsiveness of CDK4/6 inhibitors in combination with endo- crine treatment, and other markers need to be further explored and verified in clinical studies. The mechanism of action, the status of application, and mechanisms of drug-resistance of CDK4/6 inhibitors in the treatment of malignancies were reviewed and summarized in this paper, and the controversy over clinical decision-making on the treatment of breast cancer with CDK4/6 inhibitors was briefly discussed. -
利用测序技术分析肿瘤遗传变异、特异性代谢活动及进化过程,对肿瘤发生及进展机制的研究有重要意义,且为肿瘤临床治疗提供理论基础。相对于整体测序,单细胞测序技术更有利于阐释肿瘤异质性,且能从有限的样品中获得更多的信息。成人最常见的脑恶性肿瘤是脑转移瘤,而原发脑恶性肿瘤以胶质母细胞瘤(glioblastoma, GBM)最为常见,存在较强的异质性。在分子病理指导精准医疗的背景下,脑恶性肿瘤患者或因无法接受颅脑手术而失去分子病理检测和更有效的靶向治疗的机会。同时,由于颅脑手术创伤较大,多数患者无法在病情进展时重复手术治疗,利用外周血、脑脊液等微创活检的方式进行病程监测并指导个体化治疗是脑肿瘤治疗的方向。因此,鉴于单细胞测序技术的优势,其在脑恶性肿瘤临床诊疗中的应用备受期待。本文将综述单细胞测序技术流程及应用该技术进行肿瘤研究的进展,并结合脑恶性肿瘤的特征对单细胞测序技术在其诊疗中的应用进行展望。
1. 单细胞测序技术发展概述
癌细胞具有基因组不稳定性,在肿瘤起始和进展过程中可积累大量遗传突变,如单核苷酸突变、染色体变异等。利用测序技术测定癌细胞突变类型、功能,有助于探究癌症发生发展机制,并有针对性地研制治疗方案。依据DNA复制的分子生物学原理,Sanger等利用双脱氧核苷酸链终止法进行DNA片段测序,开启了基因组测序的新时代。但Sanger测序法操作繁琐、耗时长、成本高,大规模应用受限。随着荧光染色和毛细管微阵列电泳技术应用于基因组测序,第二代测序技术(next- generation sequencing,NGS)基于“边合成边测序”的原则,实现大规模、高通量测序,显著降低了测序成本。NGS法也具有一定局限性,包括测序片段较短,不能反映基因组长片段的变异,如拷贝数变异(copy number alteration,CNA)和染色体结构变异。第三代测序利用单分子实时测序及基于二代测序的合成长读长测序技术,DNA片段读长可至数十Kb,同时可进行完整的mRNA测序, 有利于对特殊基因的外显子剪切进行精确分析[1]。
既往肿瘤多组学研究通常采用NGS和/或Sanger测序技术进行肿瘤组织整体测序,获得一块肿瘤组织中成千上万个细胞的总体特征,在鉴别并筛选肿瘤相关突变基因、信号通路方面取得大量成果,促进了肿瘤临床诊断和治疗的发展。但肿瘤组织存在异质性,且不同亚型的肿瘤细胞之间的相互作用可影响疾病进程。与肿瘤组织整体测序相比,单细胞测序技术在评估肿瘤内异质性及解决特殊临床问题方面具有优势。第一,单细胞测序技术可用于分析肿瘤内异质性。第二,对于一些浸润生长的肿瘤,单细胞测序技术的应用可更高效地分析肿瘤信息。例如胰腺癌活检组织中肿瘤细胞仅占30%,其余为大量基质细胞。此时,肿瘤细胞中基因组变异可因频率过低而不能检出,尤其是CNA等变异可因测序深度不足而被忽略。第三,对于肿瘤组织样本,临床常因取样少等原因样本受限,如细针活检样本、外周血循环肿瘤细胞(circulating tumor cells,CTCs)等,不足以构建测序文库,单细胞测序或可为临床决策提供有价值的信息[2]。
2. 肿瘤单细胞测序技术概述
相对于整体测序,单细胞测序数据的获取依赖于某些技术的发展,包括单个细胞的分离及分选、单细胞全基因组测序和全外显子测序、单细胞转录组扩增及测序、单细胞表观遗传测序,以满足测序底物要求、测序成本降低和足够的测序细胞数量、控制并评估测序结果的偏倚及误差。
2.1 肿瘤单细胞分离及分选
分离及分选用于测序的单个细胞是肿瘤单细胞测序技术的重要步骤,主要包括实体肿瘤细胞、肿瘤干细胞(cancer stem cells,CSCs)、CTCs和骨髓播散肿瘤细胞(disseminated tumor cells,DTCs)[3]。其中,实体瘤可通过机械切割及胶原酶消化分散为单个细胞,而DTCs来源于骨髓穿刺。同时,该技术还可应用于肿瘤微环境中其他类型细胞的分离及分选,如免疫细胞。
CTCs是存在于患者循环系统中的肿瘤细胞,在血液中数量非常少,每毫升血液仅有数十或数百个CTCs[4],目前分离主要依据其物理或生物学特性。与循环系统中的血细胞相比,CTCs体积更大、密度小且所带电荷与表面蛋白相关,据此可利用梯度离心、微流体惯性聚焦(microfluidic inertial focusing, MIF)、微滤过器(microfabricated filters, MF)法和双向电泳法等进行分离[5]。CTCs的生物学分离方法包括免疫磁珠分离(immunomagnetic separation, IS)、经微流体免疫分离(microfluidics-enabled immunoseparation, MI)、固相酶联免疫斑点技术(epithelial immunospot, EPISPOT)法和入侵实验(invasion assay, IA)等[5]。其中最常见的是IS法,该技术利用携带捕获剂(通常为特异性抗体)的免疫磁珠富集具有特异性膜蛋白的CTCs。基于该技术建立的CellSearch检测系统已获得美国食品药品监督管理局(Food and Drug Administration,FDA)的批准[6]。考虑到测序成本及肿瘤异质性,在将样品分散成单个细胞后,有时需进一步分选用于测序的单细胞,特别是从实体肿瘤中取得的样本。样本细胞筛选通常根据CTCs的特殊分子标志或细胞表型进行,包括流式细胞术(flow cytometry,FACS)、微流体(microfluidics)系统、显微操作(micromanipulation)法和激光捕获显微切割(laser capture microdissection,LCM)[5]。
2.2 单细胞全基因组测序和全外显子测序
从单个细胞中提取的遗传物质不足以进行全基因组测序及分析,因此,通常使用多重置换扩增(multiple displacement amplilication,MDA)、PCR或二者相结合的方法进行基因扩增,如多重退火和成环循环扩增(multiple annealing and looping based amplification cycles, MALBAC)。PCR技术最先用于单细胞基因组扩增,包括选择散布在基因组中的保守序列或随机寡核苷酸序列作为扩增引物。由于基因组中不同位点与引物结合能力不同,在基因组扩增的过程中,存在大量的序列信息丢失。目前选择处于细胞周期中的四倍体细胞、简并寡核苷酸引物PCR(degenerate oligonucleotide primed-PCR,DOP-PCR)进行扩增[7],同时,选择自我复制的细胞进行测序可能导致结果存在偏倚。MDA技术利用ϕ29 DNA聚合酶和耐核酸外切酶的随机引物进行全基因组扩增,该过程无需重复温度循环,且错配率低于常规PCR[8]。MALBAC技术首先利用特殊的随机引物(包括27个氨基酸固定序列和8个氨基酸的可变序列)进行MDA,再对扩增产物进行PCR扩增[9]。有研究表明,MDA的基因组覆盖率高于DOP-PCR及MALBAC,利用该方法测序对单核苷酸位点变异(single nucleotide variants,SNV)的检出率更高;但DOP-PCR和MALBAC对基因组覆盖的均一性更强,有利于检出长度超过1 Mb的CNA[10]。
单细胞全外显子测序(single-cell whole exome sequencing,scWES)需进行外显子捕获和富集,主要用于编码基因的SNV分析。相对于scWES,单细胞全基因组测序(single-cell whole genome sequencing, scWGC)更利于CNA分析并获得非编码区信息,但由于存在基因组覆盖率的限制,SNV的检出效率较低。在乳腺癌中应用单细胞核基因组测序(nucleus genome sequencing, nuc-Seq)技术对G2/M期多倍体细胞核进行扩增和测序分析,可达到单碱基对分辨率[11]。
2.3 单细胞转录组扩增及测序
单细胞RNA测序(single-cell RNA sequencing, scRNA-Seq)包括cDNA构建、cDNA扩增和测序,其结果可显示细胞特异性表达谱和外显子转录的剪切及修饰过程,有助于分析细胞间的异质性。不同基因的转录水平差异较大,多数mRNA拷贝数较低,因此需通过扩增达到转录组测序所需的富集程度。Smart-Seq/Smart-Seq2、Phi29转录组扩增(phi29-transcrip-tome amplification,PTA)和基于半随机引物PCR的转录组扩增(semi-random primed PCR-based transcriptome amplification, STA)是常见的扩增手段。由于存在扩增偏差,通常将上述技术与特殊分子识别(unique molecular identifiers,UMIs)技术联用。UMIs可进行内参控制并定量检测基因表达。近期,在Smart-Seq2基础上使用Tn5转座酶进行cDNA富集可获得完整的转录起始位点信息,其在B淋巴细胞转录组测序中表现出较好的准确性和可重复性[12]。此外,类似于高通量二代测序技术,Drop-Seq可实现单细胞mRNA的高通量测序,该技术将单个细胞分离至纳升大小的PCR体系中,同一个细胞来源的扩增产物具有相同的序列标记,可显著提高测序效率[13]。
2.4 单细胞表观遗传测序
2.4.1 单细胞甲基化测序
基于传统的重亚硫酸盐甲基化测序过程,单细胞全基因组重亚硫酸盐测序技术的测序效果已在小鼠基因组中得到验证[14],该技术首先对单细胞DNA进行多次重亚硫酸盐转换处理,然后在DNA片段加上标记序列并进行扩增、测序。相比于传统方法,可有效减少重亚硫酸盐处理导致的DNA降解和信息损失。另一种常见的技术是简化重亚硫酸盐测序(reduced representation bisulfite sequencing, RRBS),该技术使用核酸内切酶消化DNA并富集特定的片段进行分析,在检测CpG岛方面更有优势[15]。受限于单细胞DNA含量,相较于整体样品甲基化测序,单细胞DNA甲基化测序技术的CpGs位点覆盖率较低。
2.4.2 单细胞染色体结构测序
肿瘤细胞的遗传物质不稳定,染色体结构可发生变化。基于染色质转座酶结合实验,scATAC-Seq技术利用Tn5转座酶对染色质开放区域进行测序[16],可用于细胞间异质性分析,但该方法仅针对染色质开放结构进行测序。此外,基于染色体构象捕获的全基因组高通量Hi-C可用于检测染色体的三维构象,如染色质环和拓扑结构域[17]。目前,单细胞染色体结构测序普遍具有单次测序数据较少、基因组不能全覆盖的局限性,但上述两种主要的技术目前还无可替代。单细胞核小体结构和甲基化测序技术通过GpC甲基转移酶处理,可同时获得染色质开放区域、DNA甲基化和核小体相位的序列信息,还可筛选转录因子结合位点和脱氧核糖核酸酶高敏感性位点(DNase hypersensitive sites,DHSs)等有重要意义的序列[18]。
2.4.3 单细胞非编码RNA测序
机体细胞中存在大量对细胞功能有重要调节作用的非编码RNA。全RNA测序可获得mRNA及包括长链非编码RNA(long noncoding RNAs,lncRNAs)在内的非多聚腺苷酸RNA[non-poly(A) RNA]序列信息。对于lncRNAs,可根据其序列长度、外显子数量、序列进化保守性等特点从全RNA测序结果中筛选有用信息[19]。但使用寡脱氧胸苷酸(oligo-dT)进行全RNA测序对non-poly(A) RNA的覆盖率仍有待提高。随机置换扩增测序技术(random displacement amplification sequencing,RamDA-Seq)可应用于单细胞RNA全长测序。该技术将逆转录随机置换扩增技术和部分随机引物相结合,对non-poly(A) RNA有较高的灵敏度[20]。同时,另一项研究简化了单细胞RNA测序技术的长度筛选过程,结合信息学分析可获得如microRNA、tRNA片段和小核RNA(small nucleolar RNAs,snoRNAs)等短序列测序结果,且在人类胚胎干细胞中得到验证[21]。
2.4.4 单细胞多组学共测序
单细胞基因组、转录组及表观遗传组学等多组学同时测定,可获得多维度信息,在细胞间异质性、代谢特异性分析等方面有重要意义。目前,已有多个平台可以通过细胞质与细胞核分离、基因组和转录组分离的方法,同时进行单细胞全基因组、转录组和甲基化组测序[22]。
3. 单细胞测序技术在肿瘤中的研究进展
3.1 循环肿瘤细胞
肿瘤患者外周血中存在CTCs和CTCs簇。有关肿瘤转移的研究表明,肿瘤细胞可发生上皮间质转化(epithelial-to-mesenchymal transition, EMT)并侵入血管,在循环系统中存活的部分肿瘤细胞即为CTCs,其与肿瘤转移有关[23]。此外,医源性因素也可导致CTCs。从前列腺癌患者外周血中通过微液滴系统富集CTCs并进行单细胞RNA测序,结果显示患者自身和不同患者之间的CTCs转录组具有异质性,且发现耐药性相关的雄激素受体存在基因突变和剪切变异,该过程受上调的非经典Wnt信号通路调控[24]。相比于单个CTC细胞,CTCs簇具有更强的转移能力。一项研究对结肠癌、乳腺癌、胃癌和前列腺癌原发肿瘤CTCs进行分析,结果表明后者特殊基因的CNA变异显著提高,如Myc和第10号染色体缺失的磷酸酶张力蛋白同源物基因(gene of phosphate and tension homology deleted on chromsome ten, PTEN),提示潜在的转移机制。且同一来源的CTCs之间显示出相对保守的CNA模式,同种肿瘤不同来源的CTCs之间以及原发肿瘤和CTCs之间SNV谱系存在较大差异,且SNV的发生具有更强的随机性[25]。由于存在扩增偏差和基因组测序覆盖程度的限制,CTCs的SNV研究发展需单细胞全基因组测序技术的进一步优化。
3.2 肿瘤进化及异质性
单细胞水平的研究可用于分析肿瘤内和肿瘤间的异质性,有助于肿瘤诊断、治疗,特别是对治疗过程中出现放化疗抵抗的机制研究有重要意义。利用nuc-Seq技术研究乳腺癌克隆进化可知,单倍体重排发生在肿瘤早期,在肿瘤细胞克隆扩增时染色体相对稳定;点突变则持续发生且与克隆异质性相关。同时,相比于雌激素受体阳性细胞,三阴性乳腺癌肿瘤细胞突变率更高[11]。利用肺腺癌患者肿瘤细胞进行异种移植,并对药物处理前后的异种移植肿瘤细胞进行单细胞mRNA测序, 可根据转录组将细胞分为不同的药物敏感性亚型[26]。依赖于多组学技术的发展,scTrio-Seq应用于人肝细胞肝癌基因组、甲基化组和转录组共测序,并根据SNV、DNA甲基化水平和转录组谱将被测细胞分为2个亚型,不同亚型具有不同的免疫识别潜能[27]。在单细胞基因组学和转录组学研究的基础上,表观遗传学测序结果可对基因组转录和翻译过程提供更多信息。
3.3 分子标志物筛选
既往黑色素瘤、胰腺肿瘤等相关研究提示,特征基因突变和基因重排谱是目前肿瘤诊断和预后评估的标志物之一[28-29]。针对肾透明细胞癌的单细胞外显子测序结果与既往利用整体测序技术得到的突变谱不同,且在被测序的25个细胞中未发现显著的克隆亚型[30]。另一项针对膀胱癌的研究中,对分离的膀胱癌干细胞、膀胱癌非干细胞、膀胱上皮细胞等类型的细胞进行单细胞DNA-Seq,结果显示膀胱癌干细胞存在克隆同源性,并发现了新的与肿瘤起始和细胞增殖相关的突变基因[31]。由于受到肿瘤异质性和测序成本的限制,被测序细胞的选择至关重要,因此,不仅需要发展高通量单细胞测序技术,还应建立用于单细胞检测的细胞分选标准。目前,已有研究利用单细胞RNA测序结果,构建能预测胶质瘤患者生存预后的转录组特征谱[32]。
3.4 肿瘤转移
实体肿瘤可发生远处转移和微转移。肿瘤细胞微转移DTCs通常出现在骨髓中,部分对治疗有抗性,可进展为肿瘤骨转移、内脏转移。利用单细胞测序技术可检出DTCs并定位其原发灶。从乳腺癌患者骨髓样品中分离得到单个细胞并进行全基因组测序,将序列信息中的CNA模式和原发肿瘤、淋巴结转移灶进行比对,可确定DTCs的原发肿瘤来源[33]。另一方面,对肿瘤原发灶和转移灶进行单细胞测序有助于发现肿瘤转移和定植相关的分子标志物及生态位特点。对头颈鳞癌原发灶及淋巴结转移灶进行单细胞全外显子组和转录组测序,结果显示原发灶细胞表达谱具有空间特异性,位于前缘的细胞部分上皮间质转化(partial epithelial-to-mesenchymal transition,p-EMT)水平较高,可作为肿瘤转移的预测标志[34]。
3.5 肿瘤微环境及免疫微环境
利用高通量单细胞测序技术可同时获得肿瘤微环境中的多种信息,有助于肿瘤生态位的研究。一项关于转移性黑色素瘤微环境的研究对患者肿瘤组织中的癌细胞、免疫细胞、间质细胞和内皮细胞进行单细胞RNA测序,结果显示肿瘤微环境中细胞间相互作用的特殊模式。针对肿瘤浸润T淋巴细胞的测序分析显示了该肿瘤组织中T淋巴细胞活化和克隆机制,为靶向治疗和免疫治疗提供了新思路[35]。肿瘤浸润T淋巴细胞在免疫治疗中有重要作用,从肝细胞癌患者血液、癌组织及癌旁组织中分离T淋巴细胞并进行单细胞RNA测序,可根据其分子及功能特性分型,结果显示CD8+T细胞和调节T细胞的特异性富集,测序结果亦提示了与之相关的靶基因[36]。此外,黑色素瘤中免疫细胞的测序结果显示,单核细胞和树突状细胞中干扰素γ诱导的免疫稳态调节分子的表达谱明显变化,导致抗肿瘤免疫反应被抑制,且与肿瘤免疫逃逸相关[37]。而对于脑转移瘤动物模型的单细胞RNA测序结果则提示脑肿瘤免疫微环境中小胶质细胞、骨髓来源巨噬细胞在肿瘤脑转移、脑转移瘤治疗抵抗中起重要作用[38]。
4. 脑恶性肿瘤诊治发展概述
胶质瘤约占原发脑肿瘤的27%,其中,胶质母细胞瘤是最常见的中枢神经系统原发恶性肿瘤。目前,胶质瘤的诊断主要依据影像学和组织病理学,治疗方案主要为手术结合放化疗。异柠檬酸脱氢酶(isocitrate dehydrogenase, IDH1)突变、O6-甲基鸟嘌呤-DNA甲基转移酶(O6-methylguanine-DNA methyltransferase, MGMT)甲基化、TERT启动子突变、1p19q缺失等基因组变异特点的发现是胶质瘤诊治进程中的里程碑,可指导肿瘤治疗,且与患者预后相关。世界卫生组织(World Health Organization, WHO)于2016年发布的中枢神经系统肿瘤分类指南首次将这些分子分型与组织病理整合[39]。此外,肿瘤相关抗原表皮生长因子受体(epidermal growth factorre-ceptor, EGFR)及GBM肿瘤特异性抗原EGFRvⅢ突变与胶质瘤的预后相关[40],是靶向治疗、免疫治疗(如抗原肽疫苗、过继免疫治疗等)的重要靶点。在胶质瘤的诊疗过程中,除影像学及临床进展评估外,利用血液、脑脊液中细胞学、DNA检测或可为临床进展、肿瘤复发提供辅助信息[41]。目前,胶质瘤治疗方案的优化仍需深入进行基础研究和临床试验。
脑转移瘤是最常见的中枢神经系统恶性肿瘤,发病率约为原发恶性肿瘤的3~10倍,患者预后较差。对于首诊脑转移瘤患者,手术切除及放疗是主要的治疗方法。对于原发肿瘤化疗敏感的患者,也可尝试应用传统化疗方案治疗[42]。部分患者,如EGFR突变或间变性淋巴瘤激酶重排的非小细胞肺癌、人类表皮生长因子受体2阳性的乳腺癌、BRAF突变的黑色素瘤患者,发生脑转移后可能从靶向治疗中获益。近期一项关于酪氨酸激酶抑制剂埃克替尼治疗EGFR突变的非小细胞肺癌多发脑转移的Ⅲ期临床试验(BRAIN, NCT01724801)结果显示,患者无进展生存期显著延长[43],此研究依赖于对原发肿瘤和脑转移瘤的组学研究。一项对86例对脑转移瘤及其配对原发肿瘤的单细胞测序研究表明,肿瘤发生脑转移后分子标志物发生变化,因此不能完全依赖外周肿瘤的标志物进行治疗决策,该研究亦分析了脑转移瘤在目前临床药物相关作用通路上的变异情况[44],为脑转移瘤的诊治提供了新的思路。目前关于脑转移瘤的单细胞测序研究非常有限,仍有待进一步探讨。
5. 单细胞测序技术在脑肿瘤中的研究现状
5.1 特殊分子标志及信号通路
转录因子ZEB1在EMT、DNA损伤修复、免疫抑制等多种过程中发挥重要作用,但既往关于胶质瘤中ZEB1的功能研究结果存在争议。利用单细胞测序技术测定胶质瘤组织中不同类型细胞的ZEB1分子表达,结果提示其在肿瘤细胞、反应性胶质细胞和免疫细胞之间呈不同表达模式,且与EGFR倍增、IDH1突变有关[45]。利用单细胞全基因组测序技术检测GBM中EGFR的变异谱,可发现细胞之间EGFR的CNA不同,且存在相关其他类型突变,可能是肿瘤细胞增殖、克隆变异及产生治疗抗性的机制之一[46]。此外,利用单细胞测序技术根据转录组测序结果将肿瘤组织中的细胞进行分类,为进一步分析基因和肿瘤行为的关系奠定了基础。对复发GBM患者原发肿瘤及配对复发肿瘤进行单细胞RNA测序,结果提示RAS信号通路与GBM复发相关,且该结果在一项纳入3000例胶质瘤患者肿瘤组织RNA测序结果的荟萃分析中得到证实[47]。
5.2 胶质瘤的肿瘤异质性
大量实验证实表观遗传学修饰在肿瘤形成过程中发挥重要作用。一项研究对5例原发GBM和2个GBM肿瘤干细胞系的lncRNAs表达谱进行单细胞RNA测序,结果提示肿瘤间广泛存在lncRNAs的差异表达,但存在31个lncRNAs组成的表达谱可作为肿瘤干细胞特性的分子标志物[48];此外,可根据lncRNAs表达谱将GBM细胞分为4种亚型,此分型方式与按照蛋白编码基因分型之间的一致性约为77%。对原发GBM细胞进行单细胞转录组测序,其结果显示癌基因信号、细胞增殖、补体-免疫应答及缺氧反应等代谢过程具有较强的异质性,可据此对GBM进行重新分类[49]。针对肿瘤内或肿瘤间异质性的研究可辅助治疗及预后评估。
5.3 胶质瘤的肿瘤进化
对IDH1/2突变的少突胶质瘤细胞进行单细胞RNA测序,结果显示其存在具有神经干细胞表达谱的细胞亚群,其中部分细胞高表达增殖相关基因,与全基因组CNA、点突变谱相一致,这部分细胞被认为是起始肿瘤增殖[50]。相比于整体测序,单细胞测序技术在展示肿瘤内细胞间异质性方面有较大优势,通过该技术可以更精确地分析肿瘤进化过程,并筛选有重要意义的突变形式。利用单细胞测序技术探究EGFR或PDGF突变的GBM,可以分析促血管生成因子在肿瘤发生、增殖过程中的作用,且发现PDGF受体编码基因中特定位点的缺失与前神经元性GBM增殖密切相关,提示其可能作为有意义的治疗靶点[51]。
5.4 胶质瘤的循环肿瘤细胞
目前,关于胶质瘤患者循环系统中是否存在CTCs尚存有争议,少量研究报道可在GBM患者外周血中分离得到CTCs。另一项研究针对肿瘤细胞端粒酶设计病毒载体的荧光蛋白以筛选肿瘤CTCs,利用单细胞测序技术可对该体系进行验证,实验结果提示胶质瘤患者外周血中存在CTCs,且高级别胶质瘤患者外周血中CTCs富集程度更强[52],或可将CTCs富集程度作为一种预后相关的细胞标志。
5.5 胶质瘤肿瘤微环境和免疫应答
胶质瘤存在浸润生长的特点。针对GBM患者肿瘤中心细胞、边缘浸润的肿瘤细胞及不同位置免疫细胞的单细胞测序结果表明,其基因组和转录组具有异质性,位于肿瘤中心和边缘浸润的免疫细胞之间基因表达也具有特异性。此外,不同患者之间的浸润肿瘤细胞具有相对保守的基因突变及表达模式,这些基因可能与肿瘤浸润、侵袭及GBM复发相关[53]。对IDH突变的胶质瘤细胞进行单细胞转录组测序,并结合癌症基因组图谱(the cancer genome atlas, TCGA)数据库RNA谱相关数据进行分析,该研究结果显示胶质瘤免疫微环境中小胶质细胞和巨噬细胞存在富集现象[54]。此外,肿瘤免疫微环境的进展可辅助治疗相关研究。利用单细胞RNA测序技术发现胶质瘤中存在外周血来源的巨噬细胞,高表达具有免疫抑制功能的细胞因子,且在不同级别胶质瘤中的富集程度不同[55],其可能与不同级别肿瘤恶性程度相关。
6. 未来脑恶性肿瘤诊疗应用展望
6.1 早期诊断及动态监测
目前已有部分研究报道了胶质瘤患者外周血中可分离、获得CTCs,依据胶质瘤进展、复发等过程中肿瘤组织、肿瘤干细胞及CTCs的分子标志物研究,可通过外周血中CTCs相关基因突变、表达及表观遗传学修饰提示疾病进程、治疗效果预估并监测是否复发。作为影像学检查的辅助工具,其时效性、预测的灵敏度及特异度仍有待进一步研究。CTCs富集在微创方面具有一定优势,CTCs单细胞测序或可替代组织活检,用于患者初步病理诊断并指导治疗。近期一项研究从GBM患者外周血中分离并鉴别CTCs,可作为胶质瘤复发的微创检测指标之一[56]。
此外,肺癌、乳腺癌、结直肠癌等可发生脑转移,一般预后不佳。脑转移瘤的早期检出可为治疗提供更多的选择。单细胞测序技术可对转移相关的CTCs及DTCs进行多组学测序。关于肺癌的研究发现,单细胞全基因组测序可检出CTCs中存在的特殊CNA模式,具有指示肿瘤转移的潜能[57]。此外,还可通过单细胞转录组、表观遗传组测序技术靶向CTCs或DTCs原发灶。CTCs可从患者的外周血中分离,创伤较小,且与原发肿瘤联系紧密。因此,可通过CTCs对肿瘤原发灶及转移进展的动态变化进行监测。由于CTCs在血液中富集程度较低,因此,需更高效的CTCs捕获和富集技术。为了更好地诊断和监测肿瘤,不仅需要更多关于肿瘤形成、增殖过程的多组学数据,还需对肿瘤原发灶、CTCs及肿瘤转移灶之间在分子、细胞水平上的关联和变异进行更深入的研究。
6.2 个体化治疗
单细胞测序的优势是发现肿瘤内和肿瘤间的异质性。基因组序列变异和特殊表观遗传修饰将导致细胞表达谱变化,这可能使部分细胞在治疗过程中产生耐药性,导致肿瘤细胞未被完全杀灭。利用单细胞测序技术可获得更全面的肿瘤分子特征,包括药物作用靶点、放化疗抗性标志物等,通过靶向特殊分子设计并完善治疗方案。结合针对CTCs的单细胞测序技术可实时监测肿瘤细胞异质性的变化,在治疗过程中针对其分子特征调节治疗方案。
单细胞测序技术还可对肿瘤微环境及免疫微环境进行分析,可能为临床免疫治疗提供新靶点和思路。
6.3 肿瘤分型和预后评估
相对于肿瘤组织整体测序,单细胞基因组、转录组和表观遗传组测序可获得更多特殊分子信息,结合相关分子的功能实验和临床数据,可筛选与治疗、预后相关的新型分子标志物,或针对现有标志物进行更精确的亚型分类。其次,多组学信息的整合可补充现阶段的肿瘤分子分型。此外,免疫微环境中免疫细胞的类型、数目,以及CTCs富集程度也可作为胶质瘤预后的标志物。对不同级别胶质瘤临床样本中肿瘤相关巨噬细胞(tumor-associated macrophages,TAM)进行单细胞测序分析表明,对于低级别胶质瘤,免疫微环境中高表达外周血细胞相关基因的TAM富集程度与患者预后相关[55]。
作者贡献:唐辉负责文献检索、数据分析、示意图绘制、论文撰写;应红艳参与文献检索、数据分析、论文修改;白春梅提供修改建议并帮助修改论文。利益冲突 无 -
[1] Chong QY, Kok ZH, Bui NL, et al. A unique CDK4/6 inhibitor: Current and future therapeutic strategies of abemaciclib[J]. Pharmacol Res, 2020, 156: 104686. doi: 10.1016/j.phrs.2020.104686.
[2] 国家药品监督管理局.中国上市药品目录集[EB/OL]. (2018-07-31).http://202.96.26.102/index/detail/id/511. [3] Spring LM, Wander SA, Andre F, et al. Cyclin-dependent kinase 4 and 6 inhibitors for hormone receptor-positive breast cancer: past, present, and future[J]. Lancet, 2020, 395: 817-827. DOI: 10.1016/S0140-6736(20)30165-3
[4] Hamilton E, Infante JR. Targeting CDK4/6 in patients with cancer[J]. Cancer Treat Rev, 2016, 45: 129-138. DOI: 10.1016/j.ctrv.2016.03.002
[5] Rane SG, Dubus P, Mettus RV, et al. Loss of Cdk4 expression causes insulin-deficient diabetes and Cdk4 activation results in beta-islet cell hyperplasia[J]. Nat Genet, 1999, 22: 44-52. DOI: 10.1038/8751
[6] Malumbres M, Sotillo R, Santamaría D, et al. Mammalian cells cycle without the D-type cyclin-dependent kinases Cdk4 and Cdk6[J]. Cell, 2004, 118: 493-504. DOI: 10.1016/j.cell.2004.08.002
[7] Raub TJ, Wishart GN, Kulanthaivel P, et al. Brain Exposure of Two Selective Dual CDK4 and CDK6 Inhibitors and the Antitumor Activity of CDK4 and CDK6 Inhibition in Combination with Temozolomide in an Intracranial Glioblastoma Xenograft[J]. Drug Metab Dispos, 2015, 43: 1360-1371. DOI: 10.1124/dmd.114.062745
[8] Klein ME, Kovatcheva M, Davis LE, et al. CDK4/6 Inhibitors: The Mechanism of Action May Not Be as Simple as Once Thought[J]. Cancer Cell, 2018, 34: 9-20. DOI: 10.1016/j.ccell.2018.03.023
[9] Vijayaraghavan S, Karakas C, Doostan I, et al. CDK4/6 and autophagy inhibitors synergistically induce senescence in Rb positive cytoplasmic cyclin E negative cancers[J]. Nat Commun, 2017, 8: 15916. DOI: 10.1038/ncomms15916
[10] Franco J, Balaji U, Freinkman E, et al. Metabolic Reprogramming of Pancreatic Cancer Mediated by CDK4/6 Inhibition Elicits Unique Vulnerabilities[J]. Cell Rep, 2016, 14: 979-990. DOI: 10.1016/j.celrep.2015.12.094
[11] Wang H, Nicolay BN, Chick JM, et al. The metabolic function of cyclin D3-CDK6 kinase in cancer cell survival[J]. Nature, 2017, 546: 426-430. DOI: 10.1038/nature22797
[12] Goel S, DeCristo MJ, Watt AC, et al. CDK4/6 inhibition triggers anti-tumour immunity[J]. Nature, 2017, 548: 471-475. DOI: 10.1038/nature23465
[13] Deng J, Wang ES, Jenkins RW, et al. CDK4/6 Inhibition Augments Antitumor Immunity by Enhancing T-cell Activation[J]. Cancer Discov, 2018, 8: 216-233. DOI: 10.1158/2159-8290.CD-17-0915
[14] Zhang J, Bu X, Wang H, et al. Cyclin D-CDK4 kinase destabilizes PD-L1 via cullin 3-SPOP to control cancer immune surveillance[J]. Nature, 2018, 553: 91-95. DOI: 10.1038/nature25015
[15] Hart CD, Migliaccio I, Malorni L, et al. Challenges in the management of advanced, ER-positive, HER2-negative breast cancer[J]. Nat Rev Clin Oncol, 2015, 12: 541-552. DOI: 10.1038/nrclinonc.2015.99
[16] Butt AJ, McNeil CM, Musgrove EA, et al. Downstream targets of growth factor and oestrogen signalling and endocrine resistance: the potential roles of c-Myc, cyclin D1 and cyclin E[J]. Endocr Relat Cancer, 2005, 12: S47-S59. DOI: 10.1677/erc.1.00993
[17] Sutherland RL, Musgrove EA. CDK inhibitors as potential breast cancer therapeutics: new evidence for enhanced efficacy in ER+ disease[J]. Breast Cancer Res, 2009, 12:112.
[18] Network CGA. Comprehensive molecular portraits of human breast tumours[J]. Nature, 2012, 490: 61-70. DOI: 10.1038/nature11412
[19] Finn RS, Martin M, Rugo HS, et al. Palbociclib and Letrozole in Advanced Breast Cancer[J]. N Engl J Med, 2016, 375: 1925-1936. DOI: 10.1056/NEJMoa1607303
[20] Cristofanilli M, Turner NC, Bondarenko I, et al. Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2-negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): final analysis of the multicentre, double-blind, phase 3 randomised controlled trial[J]. Lancet Oncol, 2016, 17: 425-439. DOI: 10.1016/S1470-2045(15)00613-0
[21] Slamon DJ, Neven P, Chia S, et al. Overall Survival with Ribociclib plus Fulvestrant in Advanced Breast Cancer[J]. N Engl J Med, 2020, 382: 514-524. DOI: 10.1056/NEJMoa1911149
[22] Sledge GW, Toi M, Neven P, et al. The Effect of Abemaciclib Plus Fulvestrant on Overall Survival in Hormone Receptor-Positive, ERBB2-Negative Breast Cancer That Progressed on Endocrine Therapy-MONARCH 2: A Randomized Clinical Trial[J]. JAMA Oncol, 2019, 6: 116-124. http://publikationen.uni-tuebingen.de/xmlui/handle/10900/109078
[23] Dickler MN, Tolaney SM, Rugo HS, et al. MONARCH 1, A Phase Ⅱ Study of Abemaciclib, a CDK4 and CDK6 Inhibitor, as a Single Agent, in Patients with Refractory HR+/HER2- Metastatic Breast Cancer[J]. Clin Cancer Res, 2017, 23:5218-5224. DOI: 10.1158/1078-0432.CCR-17-0754
[24] Franco Y, Ramakrishnan V, Vaidya T, et al. A systems pharmacology model to evaluate triple drug combination therapy at overcoming resistance to anti-HER2 therapy in HER2-positive breast cancer[J]. Clin Pharmacol Ther, 2020, 107: S111.
[25] Shagisultanova E, Chalasani P, Brown-Glaberman UA, et al. Tucatinib, palbociclib, and letrozole in HR+/HER2+ metastatic breast cancer: Report of phase IB safety cohort[J]. J Clin Oncol, 2019, 37: 1029. DOI: 10.1200/JCO.2019.37.15_suppl.1029
[26] Herold CI, Trippa L, Li T, et al. A phase 1b study of the CDK4/6 inhibitor ribociclib in combination with the PD-1 inhibitor spartalizumab in patients with hormone receptor-positive metastatic breast cancer (HR+ MBC) and metastatic ovarian cancer (MOC)[J]. Cancer Res, 2020, 80: SABCS19-P3-14-03.
[27] Du Q, Guo X, Wang M, et al. The application and prospect of CDK4/6 inhibitors in malignant solid tumors[J]. J Hematol Oncol, 2020, 13: 41. DOI: 10.1186/s13045-020-00880-8
[28] O'Leary B, Finn RS, Turner NC. Treating cancer with selective CDK4/6 inhibitors[J]. Nat Rev Clin Oncol, 2016, 13: 417-430. DOI: 10.1038/nrclinonc.2016.26
[29] Goldman JW, Mazieres J, Barlesi F, et al. A randomized phase 3 study of abemaciclib versus erlotinib in previously treated patients with stage Ⅳ NSCLC with KRAS mutation: JUNIPER[J]. J Clin Oncol, 2018, 36:9025. DOI: 10.1200/JCO.2018.36.15_suppl.9025
[30] Patnaik A, Rosen LS, Tolaney SM, et al. Efficacy and Safety of Abemaciclib, an Inhibitor of CDK4 and CDK6, for Patients with Breast Cancer, Non-Small Cell Lung Cancer, and Other Solid Tumors[J]. Cancer Discov, 2016, 6: 740-753. DOI: 10.1158/2159-8290.CD-16-0095
[31] Anders CK, Le Rhun E, Bachelot TD, et al. A phase Ⅱ study of abemaciclib in patients (pts) with brain metastases (BM) secondary to HR+, HER2-metastatic breast cancer (MBC)[J]. J Clin Oncol, 2019, 37: 1017. DOI: 10.1200/JCO.2019.37.15_suppl.1017
[32] Sahebjam S, Le Rhun E, Queirolo P, et al. 331P - A phase Ⅱ study of abemaciclib in patients (pts) with brain metastases (BM) secondary to non-small cell lung cancer (NSCLC) or melanoma (MEL)[J]. Ann Oncol, 2019:30. doi: 10.1093/annonc/mdz242.026.
[33] Condorelli R, Spring L, O'Shaughnessy J, et al. Polyclonal RB1 mutations and acquired resistance to CDK 4/6 inhibitors in patients with metastatic breast cancer[J]. Ann Oncol, 2018, 29: 640-645. DOI: 10.1093/annonc/mdx784
[34] Álvarez-Fernández M, Malumbres M. Mechanisms of Sensitivity and Resistance to CDK4/6 Inhibition[J]. Cancer Cell, 2020, 37: 514-529. DOI: 10.1016/j.ccell.2020.03.010
[35] McCartney A, Migliaccio I, Bonechi M, et al. Mechanisms of Resistance to CDK4/6 Inhibitors: Potential Implications and Biomarkers for Clinical Practice[J]. Front Oncol, 2019, 9: 666. DOI: 10.3389/fonc.2019.00666
[36] Pandey K, An HJ, Kim SK, et al. Molecular mechanisms of resistance to CDK4/6 inhibitors in breast cancer: A review[J]. Int J Cancer, 2019, 145: 1179-1188. DOI: 10.1002/ijc.32020
[37] Laroche-Clary A, Chaire V, Algeo MP, et al. Combined targeting of MDM2 and CDK4 is synergistic in dedifferentiated liposarcomas[J]. J Hematol Oncol, 2017, 10: 123. DOI: 10.1186/s13045-017-0482-3
[38] Herrera-Abreu MT, Palafox M, Asghar U, et al. Early Adaptation and Acquired Resistance to CDK4/6 Inhibition in Estrogen Receptor-Positive Breast Cancer[J]. Cancer Res, 2016, 76: 2301-2313. DOI: 10.1158/0008-5472.CAN-15-0728
[39] Michaloglou C, Crafter C, Siersbaek R, et al. Combined Inhibition of mTOR and CDK4/6 Is Required for Optimal Blockade of E2F Function and Long-term Growth Inhibition in Estrogen Receptor-positive Breast Cancer[J]. Mol Cancer Ther, 2018, 17: 908-920. DOI: 10.1158/1535-7163.MCT-17-0537
[40] Finn RS, Aleshin A, Slamon DJ. Targeting the cyclin-dependent kinases (CDK) 4/6 in estrogen receptor-positive breast cancers[J]. Breast Cancer Res, 2016, 18: 17. DOI: 10.1186/s13058-015-0661-5
[41] Costa C, Wang Y, Ly A, et al. PTEN Loss Mediates Clinical Cross-Resistance to CDK4/6 and PI3Kα Inhibitors in Breast Cancer[J]. Cancer Discov, 2020, 10: 72-85. http://www.researchgate.net/publication/336355817_PTEN_loss_mediates_clinical_cross-resistance_to_CDK46_and_PI3Ka_inhibitors_in_breast_cancer
[42] Yang C, Li Z, Bhatt T, et al. Acquired CDK6 amplification promotes breast cancer resistance to CDK4/6 inhibitors and loss of ER signaling and dependence[J]. Oncogene, 2017, 36: 2255-2264. DOI: 10.1038/onc.2016.379
[43] Shaulian E, Karin M. AP-1 in cell proliferation and survival[J]. Oncogene, 2001, 20: 2390-2400. DOI: 10.1038/sj.onc.1204383
[44] Teh JLF, Aplin AE. Arrested Developments: CDK4/6 Inhibitor Resistance and Alterations in the Tumor Immune Microenvironment[J]. Clin Cancer Res, 2019, 25: 921-927. DOI: 10.1158/1078-0432.CCR-18-1967
[45] Finn RS, Dering J, Conklin D, et al. PD 0332991, a selective cyclin D kinase 4/6 inhibitor, preferentially inhibits proliferation of luminal estrogen receptor-positive human breast cancer cell lines in vitro[J]. Breast Cancer Res, 2009, 11: R77. DOI: 10.1186/bcr2419
[46] Turner NC, Liu Y, Zhu Z, et al. Cyclin E1 Expression and Palbociclib Efficacy in Previously Treated Hormone Receptor-Positive Metastatic Breast Cancer[J]. J Clin Oncol, 2019, 37: 1169-1178. DOI: 10.1200/JCO.18.00925
[47] Formisano L, Lu Y, Servetto A, et al. Aberrant FGFR signaling mediates resistance to CDK4/6 inhibitors in ER+ breast cancer[J]. Nat Commun, 2019, 10: 1373. DOI: 10.1038/s41467-019-09068-2
[48] Li J, Fu F, Yu L, et al. Cyclin-dependent kinase 4 and 6 inhibitors in hormone receptor-positive, human epidermal growth factor receptor-2 negative advanced breast cancer: a meta-analysis of randomized clinical trials[J]. Breast Cancer Res Treat, 2020, 180: 21-32. DOI: 10.1007/s10549-020-05528-2
[49] André F, Ciruelos EM, Juric D, et al. Overall survival (os) results from SOLAR-1, a phase Ⅲ study of alpelisib (ALP) + fulvestrant (FUL) for hormone receptor-positive (HR+), human epidermal growth factor receptor 2-negative (HER2-) advanced breast cancer (ABC)[J]. Ann Oncol, 2020, 31: S1150-S1151. DOI: 10.1016/j.annonc.2020.08.2246
[50] Turner S, Chia SKL, Kanakamedala H, et al. Real-world effectiveness of alpelisib (ALP) + fulvestrant (FUL) compared with standard treatment among patients (Pts) with hormone-receptor positive (HR+) human epidermal growth factor receptor-2 negative (HER2-) PIK3CA-mutated (Mut) advanced breast cancer (ABC)[J]. Ann Oncol, 2020, 31: S366.
-
期刊类型引用(5)
1. 郭文杰,刘芳,高娜,陈彻,楚惠媛. 肿瘤细胞周期调控研究进展. 中国免疫学杂志. 2024(06): 1315-1319 .  百度学术
百度学术
2. 刘远亭,韩淑萍,刘沙,杨证玉,何俊超,张燕琴. 基于网络药理学探讨长春新碱治疗乳腺癌的作用机制. 食品与药品. 2023(06): 567-575 .  百度学术
百度学术
3. 姜惠心,祁子泰,高修歌,郭大伟,季辉,彭麟,江善祥. 犬乳腺肿瘤化学治疗药物的研究进展. 畜牧与兽医. 2021(12): 116-122 .  百度学术
百度学术
4. 毛倩倩,逯遥,黄彬,林久茂. 片仔癀对人结肠癌细胞HCT-116裸鼠皮下移植瘤的抑制作用. 福建中医药. 2021(12): 15-19 .  百度学术
百度学术
5. 焦秋香,陈新艳. miR-198在卵巢癌中的表达及对卵巢癌细胞增殖作用的机制研究. 标记免疫分析与临床. 2021(12): 2103-2108+2151 .  百度学术
百度学术
其他类型引用(2)

 作者投稿
作者投稿 专家审稿
专家审稿 编辑办公
编辑办公 邮件订阅
邮件订阅 RSS
RSS

 下载:
下载:











