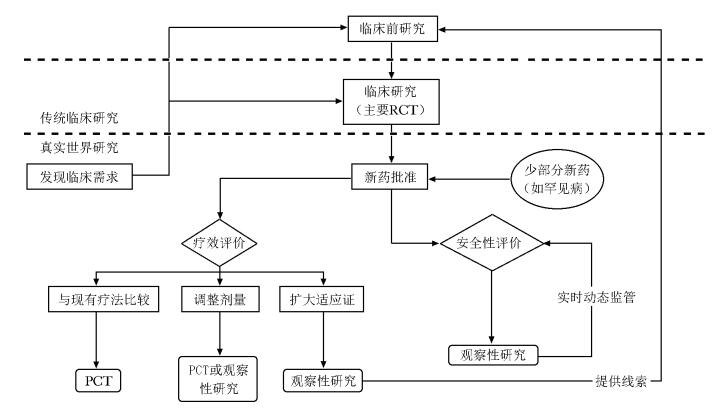
-
摘要: 真实世界证据是在现实医疗环境中, 通过优化分析来源于多种途径的数据而获得的证据。其与传统临床研究证据的本质区别在于获取数据的环境, 而非研究方法或试验设计。与传统随机对照临床试验证据相比, 真实世界证据具有结果外推性好、研究效率高等优势, 可广泛应用于医疗产品有效性和安全性评价、临床实践过程分析等领域。近年来, 随着信息技术的飞速发展和医学循证需求的持续增加, 真实世界研究愈发受到人们重视, 发展前景良好。
-
关键词:
- 真实世界证据 /
- 传统随机对照临床试验证据
Abstract: Real-world evidence is defined as information on health care that is derived from multiple sources outside typical clinical research settings. The essential difference between real-world evidence and evidence from traditionally randomized clinical trial is the setting where data is collected, but not the study method or design. Real-world evidence has a better external validity and research efficiency when compared to evidence from traditionally randomized clinical trials, and thus is widely used in the assessment of the safety and efficacy of a medical product, in the analysis of clinical practice, etc. In recent years, increasing importance has been attached to real-world research in the context of rapid development of information technology and growing need for clinical evidence, suggesting its good prospects in the future. -
表 1 真实世界研究与随机对照临床试验比较
比较项目 真实世界研究 随机对照临床试验 研究性质 效果研究, 外部效度强 效力研究, 内部效度强 研究时间 较长 较短 研究对象 无特殊限定, 符合临床实践 一般纳入特定年龄段, 无合并疾病者 样本量 大, 尽量覆盖广泛的患者人群 研究前计算具有检验效能的最小样本量 干预 按照临床实际情况给予干预, 可随机分组 随机分组后予以严格控制的干预措施, 限制合并用药等干扰因素 盲法 可以不使用 常使用 结局测量 多种, 临床意义明确 一种或几种, 常为替代指标 药品审批 主要为上市后研究 主要为上市前研究 伦理 易满足 较难满足 表 2 真实世界证据及随机对照临床试验证据的优势及不足
数据来源 优势 不足 真实世界证据 贴近临床实践, 外推性好; 证据容易获得, 研究效率高; 结局指标临床意义明确, 可反映医疗产品的实际效果及安全性; 可行亚组分析, 建立各人群的风险-获益模型 数据常不完整、准确性差, 可能与所研究问题的相关性不足; 偏倚多; 数据收集和结局测量未标准化; 样本异质性可能会掩盖疗效; 涉及隐私问题 随机对照临床试验证据 数据完整准确, 与所研究问题的相关性好; 偏倚少; 数据收集和结局测量有明确标准; 反映效力 只适用于特定人群及临床环境, 外推性差; 获取证据的效率低, 成本高; 常使用替代指标, 临床意义有待明确 -
[1] Sherman RE, Anderson SA, Dal Pan GJ, et al. Real-world evidence-what is it and what can it tell us?[J]. N Engl J Med, 2016, 375:2293-2297. doi: 10.1056/NEJMsb1609216 [2] Ford I, Norrie J. Pragmatic trials[J]. N Engl J Med, 2016, 375:454-463. doi: 10.1056/NEJMra1510059 [3] Schilsky RL. Finding the evidence in real-world evidence:moving from data to information to knowledge[J]. J Am Coll Surg, 2017, 24:1-7. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=10.1177/074171365700800106 [4] Balas EA, Boren SA. Managing clinical knowledge for health care improvement[J]. Yearb Med Inform, 2000, 1:65-70. http://www.ihi.org/resources/Pages/Publications/Managingclinicalknowledgeforhealthcareimprovement.aspx [5] Grady C, Cummings SR, Rowbotham MC, et al. Informed consent[J]. N Engl J Med, 2017, 376:856-867. doi: 10.1056/NEJMra1603773 [6] Real world evidence. Academy of Medical Sciences and Association of the British Pharmaceutical Industry[DB/OL] [2017-08-21]. https: //acmedsci.ac.uk/viewFile/56cab22108cf9.pdf. [7] Grapow MT, Wattenwyl RV, Guller U, et al. Randomized controlled trials do not reflect reality:real-world analyses are critical for treatment guidelines![J]. J Thorac Cardiovasc Surg, 2006, 132:5-7. doi: 10.1016/j.jtcvs.2006.03.035 [8] Brett W, Hirschmann MT, Guller U, et al. CABG vs. PCI in coronary artery disease:what is the evidence?[J]. Card Surg Today, 2005, 2:43-55. https://pubmed.ncbi.nlm.nih.gov/30819365/ [9] Colli A, Pagliaro L, Duca P. The ethical problem of randomization[J]. Intern Emerg Med, 2014, 9:799-804. doi: 10.1007/s11739-014-1118-z [10] 李敏, 时景璞, 于慧会.真实世界研究与随机对照试验、单病例随机对照试验在临床治疗性研究中的关系比较[J].中华流行病学杂志, 2012, 33:342-345. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zhlxbx201203021 [11] Food and Drug Administration. Use of real-world evidence to support regulatory decision-making for medical devices: Draft guidance for industry and Food and Drug Administration staff[DB/OL].[2016]. http://www.fda.gov/downloads/MedicalDevices/DeviceRegulationandGuidance/GuidanceDocuments/UCM513027.pdf. [12] Kremers HM. Methods to analyze real-world databases and registries[J]. Bull NYU Hosp Jt Dis, 2009, 67:193-197. http://cn.bing.com/academic/profile?id=e17e740a87aeb68dd2fa40efabf0f0ab&encoded=0&v=paper_preview&mkt=zh-cn [13] Tunis SR, Stryer DB, Clancy CM. Practical clinical trials increasing the value of clinical research for decision making in clinical and health policy[J]. JAMA, 2003, 290:1624-1632. [14] Aspirin dosing: a patient-centric trial assessing benefits and long-term(ADAPTABLE).[2017-08-21]. https: //clinicaltrials.gov/ct2/show/NCT02697916. [15] Peikes D, Geonnotti K, Wang W. Using pragmatic clinical trials to test the effectiveness of patient-centered medical home models in real-world settings(issue brief)[DB/OL]. Mathematica Policy Research, 2013. https: //pcmh.ahrq.gov/sites/default/files/attachments/UsingPragmatic_032513comp.pdf. [16] Roberts I, Yates D, Sandercock P, et al. Effect of intravenous corticosteroids on death within 14 days in 10008 adults with clinically significant head injury(MRC CRASH trial):randomised placebo-controlled trial[J]. Lancet, 2004, 364:1321-1328. doi: 10.1016/S0140-6736(04)17188-2 [17] Campbell R, Starkey F, Holliday J, et al. An informal school-based peer-led intervention for smoking prevention in adolescence(ASSIST):a cluster randomised trial[J]. Lancet, 2008, 371:1595-1602. doi: 10.1016/S0140-6736(08)60692-3 [18] Bhatt DL, Mehta C. Adaptive designs for clinical trials[J]. N Engl J Med, 2016, 375:65-74. doi: 10.1056/NEJMra1510061 [19] Donegan K, King B, Bryan P. Safety of pertussis vaccination in pregnant women in UK:observational study[J]. BMJ, 2014, 349:g4219. doi: 10.1136/bmj.g4219 [20] Rupprecht CE, Briggs D, Brown CM, et al. Use of a reduced(4-dose) vaccine schedule for postexposure prophylaxis to prevent human rabies:recommendations of the advisory committee on immunization practices[J]. MMWR Recomm Rep, 2010, 59:1-9. https://www.cdc.gov/rabies/resources/acip_recommendations.html [21] National Academies of Sciences, Engineering, and Medicine(US); Health and Medicine Division(US); Board on Health Sciences Policy(US); Forum on Drug Discovery, Development, and Translation(US). Real-world evidence generation and evaluation of therapeutics: proceedings of a workshop[M/OL]. Washington, DC: National Academies Press, 2017. https: //www.ncbi.nlm.nih.gov/books/NBK424402/doi: 10.17226/24685. [22] Taylor H, Burcombe R, Hill S, et al. Assessing the impact on staff resource and patient waiting time of a switch from IV to oral chemotherapy:time and motion model for HTAs[J]. Value Health, 2005, 8:A45. http://www.sciencedirect.com/science/article/pii/S1098301510672762 [23] 孙鑫, 谭婧, 康力, 等.重新认识真实世界研究[J].中国循证医学杂志, 2017, 17:0-4. [24] Li G, Sajobi TT, Menon BK, et al. Registry-based randomized controlled trials:advantages, challenges and areas for future research[J]. J Clin Epidemiol, 2016, 80:16-24. doi: 10.1016/j.jclinepi.2016.08.003 [25] Meldrum M. A calculated risk:the Salk polio vaccine field trials[J]. BMJ, 1998, 317:1233-1236. doi: 10.1136/bmj.317.7167.1233 -


 作者投稿
作者投稿 专家审稿
专家审稿 编辑办公
编辑办公 邮件订阅
邮件订阅 RSS
RSS


 下载:
下载: