The Prevalence and Clinical Usefulness of Anti-PUF60 Autoantibody in Patients with Idiopathic Inflammatory Myopathy
-
摘要:
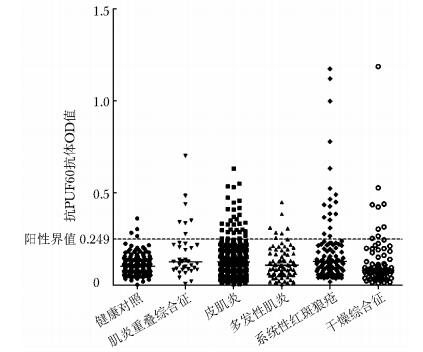
目的 检测抗PUF60抗体在中国特发性炎性肌病(idiopathic inflammatory myopathy, IIM)患者中的阳性率, 并探索其临床意义。 方法 选取1989年10月至2016年8月于中日友好医院住院治疗的388例IIM患者[275例皮肌炎(dermatomyositis, DM)、76例多发性肌炎(polymyositis, PM)及37例肌炎重叠综合征], 211例其他自身免疫性疾病患者[28例神经肌肉疾病(noninflammatory neuromuscular disease, NMD)、104例系统性红斑狼疮(systemic lupus erythematosus, SLE)及79例干燥综合征(Sj gren syndrome, SS)]作为疾病对照组; 同时选取门诊体检的167例健康成年人作为健康对照组(healthy controls, HCs)。采用酶联免疫吸附法(enzyme-linked immunosorbent assay, ELISA)检测入组患者及HCs的抗体滴度, 并联合免疫印迹实验验证ELISA结果的可靠性。组间比较用独立Mann-Wh itney U检验, 相关性分析采用Spearman秩检验。 结果 抗PUF60抗体可见于11.6%(45/388)的IIM患者, 17.3%(18/104)的SLE患者, 10.1%(8/79)的SS患者, 阳性率均高于HCs(IIM比HCs, P < 0.001;SLE比HCs, P < 0.001;SS比HCs, P=0.009)。且该抗体可见于各IIM亚组, 包括11.3%(31/275)的DM患者和21.6%(8/37)的肌炎重叠综合征患者, 阳性率均高于HCs(DM比HCs, P < 0.001;肌炎重叠综合征比HCs, P < 0.001)。此外, 抗PUF60抗体在不同自身免疫性疾病中的临床相关性各异。在经典DM中, 该抗体常见于肌炎抗体阴性(P=0.018)及并发技工手(P=0.046)的患者; 而且该抗体在无肌病性皮肌炎中的阳性率较高, 且多伴发皮肤溃疡(P=0.029)。在其他自身免疫性疾病中发现, 抗体阳性的SS患者皮疹发生率及类风湿因子、抗SSB抗体、抗RNP抗体阳性比例增加, 且抗体阳性的SLE患者外周血CD5+CD19+B细胞数目升高及抗双链DNA、抗Sm、抗SSB抗体阳性比例同样增加(P均 < 0.05)。 结论 抗PUF60抗体可见于中国DM、肌炎重叠综合征、SLE和SS患者, 且在不同类型自身免疫性疾病中的临床相关性各异, 提示抗PUF60抗体或其靶抗原PUF60可能在不同自身免疫性疾病中参与了不同致病过程。 Abstract:Objective The aim of our study was to detect the positive incidence and analyze the clinical significance of anti-PUF60 antibody in Chinese patients with idiopathic inflammatory myopathy(IIM). Methods This study involved, from October 1989 to August 2016, 388 IIM patients (including 275 patients with dermatomyositis(DM), 76 patients with polymyositis(PM), and 37 patients with myositis overlap syndrome), 211 patients with control diseases (including 28 patients with noninflammatory neuromuscular disease(NMD), 104 patients with systemic lupus erythematosus(SLE), and 79 patients with Sj gren's syndrome(SS)) as disease controls, and 167 healthy adults as healthy controls(HCs). Enzyme-linked immunosorbent assay(ELISA) was developed to detect serum anti-PUF60 antibody, and immunoblotting analyses were performed to validate the ELISA results. The results of two groups were compared using unpaired Mann-Whitney U test, and the relevance was analyzed using Spearman correlation analysis. Results Anti-PUF60 antibody was detected in 11.6%(45/388) of patients with IIM, 17.3%(18/104) of patients with SLE, and 10.1%(8/79) of patients with SS, and the antibody positive rates were significantly higher than that of HCs(P < 0.001, P < 0.001, and P=0.009, respectively). In IIM subgroups, anti-PUF60 antibody presented in 11.3%(31/275) of DM patients and 21.6%(8/37) of patients with myositis overlap syndrome, and the antibody positive rates were significantly higher than that of HCs(P=0.001 and P < 0.001, respectively). The clinical associations of anti-PUF60 antibody in different groups of immune disease were varied. Classical DM patients with anti-PUF60 antibody had lower prevalence of currently known myositis antibodies(P=0.018) and higher prevalence of mechanic's hands(P=0.046). While, the prevalence of anti-PUF60 antibody was particularly higher in the clinically amyopathic dermatomyositisc(CADM), subgroup and associated with the incidence of skin ulcer(P=0.029). As for other rheumatic diseases, in SS patients, anti-PUF60 antibody was significantly associated with higher prevalence of cutaneous manifestation, rheumatoid factor, and antibodies against SSB and RNP; in SLE patients, it is related to higher incidence of younger onset, alopecia, hypocomplementemia, elevated CD5+CD19+B-cell subpopulations, and antibodies against dsDNA, Sm and SSB. Conclusions Anti-PUF60 antibody can be detected in Chinese patients with DM, myositis-overlap syndrome, SLE and SS; and it is associated with distinct clinical features in different rheumatic diseases. This finding indicates that anti-PUF60 antibody is associated with different pathogenesis processes in different systemic rheumatic diseases. -
Key words:
- idiopathic inflammatory myopathies /
- anti-PUF60 autoantibody /
- splicesome
-
表 1 DM患者中抗PUF60抗体的临床意义
临床特点 经典DM CADM PUF60阳性(n=22) PUF60阴性(n=212) P值 PUF60阳性(n=9) PUF60阴性(n=32) P值 性别[女性(%)] 16(72.7) 147(70.3*) 0.766 7(77.8) 25(78.1) 0.982 年龄[岁, M(Q)] 52(31~58) 51(42~59) 0.561 60(49~72) 45(39~57) 0.008 发病年龄[岁, M(Q)] 42(27~55) 49(41~57) 0.151 58(48~72) 43(35~56) 0.007 发热[n(%)] 3(13.6) 49(23.5*) 0.290 1(11.1) 1(3.1) 0.395 肌无力[n(%)] 16(72.7) 140(67.3*) 0.584 - - - 肌痛[n(%)] 15(68.2) 105(50.5*) 0.109 - - - 向阳疹[n(%)] 16(72.7) 149(71.6*) 0.968 5(55.6) 20(62.5) 0.706 Gottrons征[n(%)] 11(50.0) 130(62.5*) 0.252 8(88.9) 17(53.1) 0.052 技工手[n(%)] 10(45.5) 53(25.5*) 0.046 3(33.3) 14(43.8) 0.575 雷诺现象[n(%)] 0(0) 21(10.0*) 0.118 1(11.1) 4(12.5) 0.910 吞咽困难[n(%)] 6(27.3) 72(34.6*) 0.489 - - - 皮肤溃疡[n(%)] 3(13.6) 18(8.7*) 0.445 3(33.3) 2(6.3) 0.028 心脏病[n(%)] 2(9.1) 12(5.8*) 0.535 2(22.2) 0(0) 0.044 关节炎[n(%)] 9(40.9) 60(28.8*) 0.240 4(44.4) 10(31.3) 0.461 肺间质病变[n(%)] 13(59.1) 133(63.9*) 0.737 7(77.8) 19(59.4) 0.311 恶性肿瘤[n(%)] 4(18.2) 38(17.9*) 0.976 0(0) 7(21.9) 0.123 抗TIF-1γ抗体阳性[n(%)] 2(9.5*) 52(27.4*) 0.087 2(22.2) 8(25.0) 0.864 抗PL-7抗体阳性[n(%)] 3(14.2*) 6(3.0*) 0.045 0(0) 2(6.3) 1.000 肌炎抗体阴性[n(%)] 9(45.0*) 42(21.0*) 0.018 6(75.0*) 21(65.6*) 0.613 ESR > 20mm/h[n(%)] 6(28.6*) 81(41.5*) 0.235 5(62.5*) 6(18.7) 0.013 ANA阳性[n(%)] 12(54.5*) 104(57.8*) 0.772 4(50.0*) 6(23.1*) 0.144 ASO阳性[n(%)] 2(13.3*) 1(0.6*) 0.021 - - - CD5+CD19+B细胞百分比升高[n(%)] 18(94.7*) 113(72.9*) 0.040 6(75.0*) 17(85.0*) 0.533 Th/Ts升高[n(%)] 4(22.2*) 61(37.0*) 0.202 5(62.5*) 3(14.3*) 0.007 DM:皮肌炎;CADM:临床无肌病性皮肌炎;TIF- 1γ:转录中介因子- 1γ;ESR:红细胞沉降率;ANA:抗核抗体;ASO:抗链球菌素;Th/Ts:辅助型T细胞/抑制型T细胞;*部分患者资料缺失 表 2 抗PUF60抗体阳性SLE和SS患者的临床特点
临床特点 SLE SS PUF60阳性(n=18) PUF60阴性(n=86) P值 PUF60阳性(n=8) PUF60阴性(n=71) P值 性别[女性(%)] 17(94.4) 77(89.5) 0.521 8(100) 67(94.3) 1.000 年龄[岁, M(Q)] 25(21~31) 34(25~49) 0.004 50(44~61) 58(48~66) 0.265 发病年龄[岁, M(Q)] 21(18~27) 29(20~37) 0.007 46(42~51) 49(37~57) 0.721 发热[n(%)] 6(33.3) 21(24.4) 0.433 0(0) 1(1.4) 1.000 脱发[n(%)] 13(72.2) 35(40.7) 0.015 - - - 皮肤受累[n(%)] 12(66.7) 48(55.8) 0.397 5(62.5) 8(11.3) 0.000 关节炎[n(%)] 4(22.2) 32(37.2) 0.224 1(12.5) 24(33.8) 0.219 C4下降[n(%)] 18(100) 64(78.0*) 0.028 5(62.5) 24(36.4*) 0.143 CD5+CD19+B细胞升高[n(%)] 8(66.7*) 22(33.8*) 0.032 3(60.0*) 36(80.0*) 0.306 抗dsDNA抗体阳性[n(%)] 12(85.8*) 39(54.9*) 0.032 1(20.0*) 7(11.7*) 0.493 抗Sm抗体阳性[n(%)] 8(44.4) 8(10.1*) 0.001 0(0) 1(1.6) 1.000 抗SSB抗体阳性[n(%)] 5(27.8) 4(5.1*) 0.003 6(85.7*) 14(22.2*) 0.000 抗RNP抗体阳性[n(%)] 7(38.9) 29(38.2*) 0.923 3(42.9*) 5(7.9*) 0.028 SLE:系统性红斑狼疮;SS:干燥综合征; *部分患者资料缺失 -
[1] Lazarou IN, Guerne PA. Classification, diagnosis, and management of idiopathic inflammatory myopathies[J]. J Rheumatol, 2013, 40:550-564. doi: 10.3899/jrheum.120682 [2] Tieu J, Lundberg IE, Limaye V. Idiopathic inflammatory myositis[J]. Best Pract Res Clin Rheumatol, 2016, 30:149-168. http://cn.bing.com/academic/profile?id=477acfd94e06f59f9da99499def3e074&encoded=0&v=paper_preview&mkt=zh-cn [3] Hastings ML, Allemand E, Duelli DM, et al. Control of pre-mRNA splicing by the general splicing factors PUF60 and U2AF(65)[J]. PLoS One, 2007, 2:e538. doi: 10.1371/journal.pone.0000538 [4] Fiorentino DF, Presby M, Baer AN, et al. PUF60:a prominent new target of the autoimmune response in dermatomyositis and Sjogren's syndrome[J]. Annals Rheum Dis, 2016, 75:1145-1151. doi: 10.1136/annrheumdis-2015-207509 [5] Ioannou Y, Sultan S, Isenberg DA. Myositis overlap syndromes[J]. Curr Opin Rheumatol, 1999, 11:468-474. doi: 10.1097/00002281-199911000-00004 [6] Bailey EE, Fiorentino DF. Amyopathic dermatomyositis:definitions, diagnosis, and management[J]. Curr Rheumatol Rep, 2014, 16:465. doi: 10.1007/s11926-014-0465-0 [7] Tan EM, Cohen AS, Fries JF, et al. The 1982 revised criteria for the classification of systemic lupus erythematosus[J]. Arthritis Rheum, 1982, 25:1271-1277. doi: 10.1002/art.1780251101 [8] Rischmueller M, Tieu J, Lester S. Primary Sjogren's syndrome[J]. Best Pract Res Clin Rheumatol, 2016, 30:189-220. http://cn.bing.com/academic/profile?id=1d5037fcda396eaca0df0265bb2c107d&encoded=0&v=paper_preview&mkt=zh-cn [9] Kobayashi S, Hoshino T, Hiwasa T, et al. Anti-FIRs(PUF60) auto-antibodies are detected in the sera of early-stage colon cancer patients[J]. Oncotarget, 2016, 7:82493-82503. doi: 10.18632/oncotarget.12696 [10] Matsushita K, Tomonaga T, Shimada H, et al. An essential role of alternative splicing of c-myc suppressor FUSE-binding protein-interacting repressor in carcinogenesis[J]. Cancer Res, 2006, 66:1409-1417. doi: 10.1158/0008-5472.CAN-04-4459 [11] Corsini L, Hothorn M, Stier G, et al. Dimerization and protein binding specificity of the U2AF homology motif of the splicing factor Puf60[J]. J Biol Chem, 2009, 284:630-639. doi: 10.1074/jbc.M805395200 [12] Hogg JR, Collins K. Human Y5 RNA specializes a Ro ribonucleoprotein for 5S ribosomal RNA quality control[J]. Genes Dev, 2007, 21:3067-3072. doi: 10.1101/gad.1603907 [13] Verhagen AP, Pruijn GJ. Are the Ro RNP-associated Y RNAs concealing microRNAs? Y RNA-derived miRNAs may be involved in autoimmunity[J]. Bioessays, 2011, 33:674-682. doi: 10.1002/bies.201100048 [14] Bouffard P, Barbar E, Briere F, et al. Interaction cloning and characterization of RoBPI, a novel protein binding to human Ro ribonucleoproteins[J]. RNA, 2000, 6:66-78. doi: 10.1017/S1355838200990277 [15] Mahler M, Miller FW, Fritzler MJ. Idiopathic inflammatory myopathies and the anti-synthetase syndrome:a comprehensive review[J]. Autoimmun Rev, 2014, 13:367-371. doi: 10.1016/j.autrev.2014.01.022 -


 作者投稿
作者投稿 专家审稿
专家审稿 编辑办公
编辑办公 邮件订阅
邮件订阅 RSS
RSS


 下载:
下载: