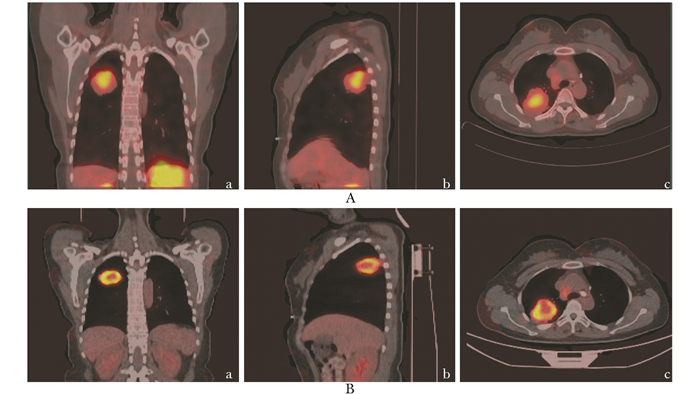
Integrin Receptor Imaging 99mTc-3PRGD2 SPECT/CT in Diagnosis and Lymph Node Staging of Non-Small Cell Lung Cancer
-
摘要:
目的 通过与18F-FDG PET/CT比较, 前瞻性评估整合素αvβ3受体显像99mTc-3PRGD2 SPECT/CT对非小细胞肺癌诊断及淋巴结分期的价值。 方法 前瞻性纳入2011年2月至2012年12月北京协和医院知情同意参与研究的65例肺占位病变患者。其中男41例, 女24例, 平均年龄(60±11)岁。患者在1周内分别行99mTc-3PRGD2 SPECT/CT和18F-FDG PET/CT检查。完成病理诊断后, 随访至死亡或随访至少2年。用受试者工作特征曲线、Z检验、卡方检验比较两种方法对肺内病灶及纵隔淋巴结的诊断效能。 结果 53例患者共65个病灶经病理证实为非小细胞肺癌, 12例患者共14个病灶诊断为良性病变。基于美国癌症联合委员会/国际抗癌联盟纵隔分区, 248个区域有肿瘤转移淋巴结, 56个区域有良性淋巴结。在诊断纵隔淋巴结转移方面, 99mTc-3PRGD2 SPECT/CT特异性显著高于18F-FDG PET/CT(94.6%比75.0%, P=0.008), 而两者的敏感性差异无统计学意义(88.3%比90.7%, P=0.557)。两种方法对肺内病变的诊断效能差异无统计学意义(Z=0.82, P=0.410)。 结论 整合素受体显像99mTc-3PRGD2 SPECT/CT在诊断淋巴结转移方面具有较高的特异性, 可能弥补18F-FDG PET/CT的不足, 在肺癌手术决策方面具有更大价值。 -
关键词:
- 整合素αvβ3 /
- 单光子发射计算机断层成像术 /
- 非小细胞肺癌 /
- 淋巴结转移
Abstract:Objective To prospectively evaluate the value of integrin αvβ3 receptor imaging approach basedon single photon emission computed tomography/computed tomography (SPECT/CT), using 99mTc-3PRGD2 as the tracer, in diagnosis and staging of non-small cell lung cancer (NSCLC) compared with 18F-FDG positron emisson tomography/computed tomography(PET/CT). Methods From February 2011 to December 2012, 65 patients with suspicious lung lesions were recruited with informed consent in Peking Union Medical College Hospital, including 41 males and 24 females, with a mean age of (60±11) years. The patients underwent both 99mTc-3PRGD2 SPECT/CT and 18F-FDG PET/CT within one week. After pathological diagnosis, all the patients were followed up until death or for at least two years. Receiver operating characteristic (ROC) curve, Z test, and Chi-square test were used to compare the diagnostic performance of the two imaging methods in assessing lung lesions and mediastinal lymph nodes. Results Sixty-five lung lesions in 53 patients were pathologically diagnosed as NSCLC, and 14 lung lesions in 12 patients were proved as benign. Based on the American Joint Committee on Cancer/Union for International Cancer Control(AJCC-UICC) standard, 248 regions of lymph nodes had metastasis and 56 were negative regions. 99mTc-3PRGD2 SPECT/CT was found to have higher specificity compared with 18F-FDG PET/CT in the per-region diagnosis of lymph node metastasis (94.6% vs. 75.0%, P=0.008), whereas the sensitivity of the two methods showed no statistically significant difference (88.3% vs. 90.7%, P=0.557). There was no significant difference between the two methods in diagnostic performance for lung lesion (Z=0.82, P=0.410). Conclusion 99mTc-3PRGD2 SPECT/CT shows high specificity in the diagnosis of lymph node metastasis from NSCLC, which may complement 18F-FDG PET/CT in diagnosis and benefit surgical decision-making for patients with lung cancer. -
表 1 99mTc-3PRGD2 SPECT/CT和18F-FDG PET/CT在肺癌诊断和转移淋巴结评估中的受试者工作特征曲线分析结果
项目 检查方法 曲线下面积 标准误 95%置信区间 Z P 肺癌诊断(n=79) 99mTc-3PRGD2 SPECT/CT 0.674 0.107 0.467~0.881 0.82 0.410 18F-FDG PET/CT 0.768 0.076 0.620~0.917 转移淋巴结评估(n=304) 99mTc-3PRGD2 SPECT/CT 0.970 0.009 0.952~0.988 8.00 <0.001 18F-FDG PET/CT 0.792 0.035 0.724~0.859 表 2 99mTc-3PRGD2 SPECT/CT和18F-FDG PET/CT在肺癌诊断和转移淋巴结评估中诊断参数的比较
项目 检查方法 阳性标准 敏感性 特异性 准确性 阳性预测值 阴性预测值 肺癌诊断 99mTc-3PRGD2 SPECT/CT T/B>4.4 64.6%(42/65) 64.3%(9/14) 64.6%(51/79) 89.4%(42/47) 28.1%(9/32) (n=79) 18F-FDG PET/CT T/B>8.0 76.9%(50/65) 64.3%(9/14) 74.7%(59/79) 90.9%(50/55) 37.5%(9/24) P值 0.039 0.625 0.210 0.793 0.457 淋巴结评估 99mTc-3PRGD2 SPECT/CT T/B>1.2 88.3%(219/248) 94.6%(53/56) 89.4%(272/304) 98.6%(219/222) 64.6%(53/82) (n=304) 18F-FDG PET/CT T/B>1.6 90.7 %(225/248) 75.0%(42/56) 87.8%(267/304) 94.1%(225/239) 64.6%(42/65) P值 0.557 0.008 0.362 0.010 0.998 -
[1] Jemal A, Bray F, Center MM, et al. Global cancer statistics[J]. CA Cancer J Clin, 2011, 61:69-90. [2] Giaccone G. 18Fluorodeoxyglucose positron emission tomography, a standard diagnostic tool in lung cancer[J]. J Natl Cancer Inst, 2007, 99:1741-1743. doi: 10.1093/jnci/djm245 [3] Fischer B, Lassen U, Mortensen J, et al. Preoperative staging of lung cancer with combined PET-CT[J]. N Engl J Med, 2009, 361:32-39. doi: 10.1056/NEJMoa0900043 [4] Vansteenkiste J, Fischer BM, Dooms C, et al. Positron-emission tomography in prognostic and therapeutic assessment of lung cancer:systematic review[J]. Lancet Oncol, 2004, 5:531-540. doi: 10.1016/S1470-2045(04)01564-5 [5] Jia B, Liu Z, Zhu Z, et al. Blood clearance kinetics, biodistribution, and radiation dosimetry of a kit-formulated integrin αvβ3-selective radiotracer 99mTc-3PRGD2 in non-human primates[J]. Mol Imaging Biol, 2011, 13:730-736. doi: 10.1007/s11307-010-0385-y [6] Zhu Z, Miao W, Li Q, et al. 99mTc-3PRGD2 for integrin receptor imaging of lung cancer:a multicenter study[J]. J Nucl Med, 2012, 53:716-722. doi: 10.2967/jnumed.111.098988 [7] Plow EF, Haas TA, Zhang L, et al. Ligand binding to integrins[J]. J Biol Chem, 2000, 275:21785-21788. doi: 10.1074/jbc.R000003200 [8] Haubner R, Weber WA, Beer AJ, et al. Noninvasive visualization of the activated αⅤβ3 integrin in cancer patients by positron emission tomography and[18F]Galacto-RGD[J]. PLOS Med, 2005, 2:244-252. doi: 10.1371/journal.pmed.0020244 [9] Mittra ES, Goris ML, Iagaru AH, et al. Pilot pharmacokine-tic and dosimetric studies of (18)F-FPPRGD2:a PET radiopharmaceutical agent for imaging α(v)β(3) integrin levels[J]. Radiology, 2011, 260:182-191. doi: 10.1148/radiol.11101139 [10] Iagaru A, Mosci C, Shen B, et al. (18)F-FPPRGD2 PET/CT:pilot phase evaluation of breast cancer patients[J]. Radiology, 2014, 273:549-559. doi: 10.1148/radiol.14140028 [11] McParland BJ, Miller MP, Spinks TJ, et al. The biodistribution and radiation dosimetry of the Arg-Gly-Asp peptide 18F-AH111585 in healthy volunteers[J]. J Nucl Med, 2008, 49:1664-1667. doi: 10.2967/jnumed.108.052126 [12] Doss M, Kolb HC, Zhang JJ, et al. Biodistribution and radiation dosimetry of the integrin marker 18F-RGD-K5 determined from whole-body PET/CT in monkeys and humans[J]. J Nucl Med, 2012, 53:787-795. doi: 10.2967/jnumed.111.088955 [13] Wan W, Guo N, Pan D, et al. First experience of 18F-alfatide in lung cancer patients using a new lyophilized kit for rapid radiofluorination[J]. J Nucl Med, 2013, 54:691-698. doi: 10.2967/jnumed.112.113563 [14] Bach-Gansmo T, Danielsson R, Saracco A, et al. Integrin receptor imaging of breast cancer:a proof-of-concept study to evaluate 99mTc-NC100692[J]. J Nucl Med, 2006, 47:1434-1439. [15] Shi J, Wang L, Kim YS, et al. Improving tumor uptake and excretion kinetics of 99mTc-labeled cyclic arginine-glycine-aspartic (RGD) dimers with triglycine linkers[J]. J Med Chem, 2008, 51:7980-7990. doi: 10.1021/jm801134k [16] Liu Z, Jia B, Shi J, et al. Tumor uptake of the RGD dimeric probe 99mTc-G3-2P4-RGD2 is correlated with integrin αvβ3 expressed on both tumor cells and neovasculature[J]. Bioconjug Chem, 2010, 21:548-555. doi: 10.1021/bc900547d [17] Wolfenson H, Iskratsch T, Sheetz MP. Early events in cell spreading as a model for quantitative analysis of biomechanical events[J]. Biophys J, 2014, 107:2508-2514. doi: 10.1016/j.bpj.2014.10.041 [18] van den Hoogen C, van der Horst G, Cheung H, et al. Integrin αv expression is required for the acquisition of a metastatic stem/progenitor cell phenotype in human prostate cancer[J]. Am J Pathol, 2011, 179:2559-2568. doi: 10.1016/j.ajpath.2011.07.011 [19] Jin X, Meng Y, Zhu Z, et al. Elevated 99mTc-3PRGD2 activity in benign metastasizing leiomyoma[J]. Clin Nucl Med, 2013, 38:117-119. doi: 10.1097/RLU.0b013e318279f14d [20] Deppen S, Putnam JB Jr, Andrade G, et al. Accuracy of FDG-PET to diagnose lung cancer in a region of endemic granulomatous disease[J]. Ann Thorac Surg, 2011, 92:428-433. doi: 10.1016/j.athoracsur.2011.02.052 [21] Miller JC, Pien HH, Sahani D, et al. Imaging angiogenesis:applications and potential for drug development[J]. J Natl Cancer Inst, 2005, 97:172-187. doi: 10.1093/jnci/dji023 -


 作者投稿
作者投稿 专家审稿
专家审稿 编辑办公
编辑办公 邮件订阅
邮件订阅 RSS
RSS


 下载:
下载: