Performance Evaluation of Clinical Detection of Coagulation Factors, Protein S, Protein C, Anti-thrombin Ⅲ, and von Willebrand Factor Antigen
-
摘要:
目的 对血栓与止血特殊项目的临床检测进行全面性能评价, 以期为临床提供可靠的检测结果。 方法 按照美国临床实验室标准化协会(Clinical and Laboratory Standards Institute, CLSI)EP5-A2、EP9-A2文件及《卫生部临床血液学检验常规项目分析质量要求WS/T 406-2012文件》标准, 对本实验室ACL TOP 700全自动凝血分析仪测定凝血因子(FⅡ、FⅤ、FⅦ、FⅧ、FⅨ、FⅩ、FⅪ、FⅫ)、易栓症筛查三项(蛋白S、蛋白C及抗凝血酶-Ⅲ)和血管性血友病因子抗原(von Willebrand factor antigen, vWF:Ag)共12个项目的准确度、不精密度、携带污染率、线性、方法学比对及参考范围进行全面性能评估。 结果 美国病理学家协会提供的12个项目质评物检测结果与靶值相比, 除蛋白C低值外偏差均小于15%。8个凝血因子批内不精密度为1.7%~4.4%, 均小于4.5%;批间不精密度为2.2%~7.5%, 均不大于7.5%。易栓症筛查三项的批内不精密度为1.0%~7.0%;批间不精密度为1.5%~10.5%。vWF:Ag批内不精密度为2.2%~3.1%, 批间不精密度为2.1%~4.1%。12个项目携带污染率为0~2.63%, 均小于3%。线性验证实验结果显示蛋白S、蛋白C、抗凝血酶-Ⅲ及vWF:Ag相关系数为0.982~0.988, 均≥0.975。ACL TOP 700与参比检测系统进行比较, 12个项目检测结果的相关系数均≥0.975, 一致性较好。厂商提供的参考范围均适用于本实验室。 结论 ACL TOP 700全自动凝血分析仪对血栓与止血特殊项目检测的准确度、不精密度、携带污染率、现行线性范围及参考范围验证等性能评价良好, 适用于临床标本检测。 Abstract:Objective To conduct a comprehensive performance evaluation of fully automated coagulation analyzer in testing coagulation factors, protein S(PS), protein C(PC), anti-thrombin Ⅲ (AT-Ⅲ), and von Willebrand factor antigen (vWF:Ag). Methods According to the Clinical and Laboratory Standards Institute (CLSI) EP5-A2, EP9-A2, and WS/T 406-2012 specifications, coagulation factors(FⅡ, FⅤ, FⅦ, FⅧ, FⅨ, FⅩ, FⅪ, and FⅫ), PS, PC, AT-Ⅲ, and vWF:Ag were detected in the apparatus to evaluate the accuracy, within-run and between-run imprecisions, linear range, carryover rate, method comparisons, and reference range of the ACL TOP 700 coagulation analyzer. Results The instrument had high accuracy and precision, a good linear range, and low carryover rate (0-2.63%, < 3%). The biases of the 12 College of American Pathologists (CAP) controls compared with the target value were all less than 15%, excepting the low value of PC. The within-run imprecisions of 8 coagulation factors were 1.7%-4.4%, and the between-run imprecisions were 2.2%-7.5%. The within-run imprecisions of 3 thrombophilia screen tests and vWF:Ag were 1.0%-7.0% and 2.2%-3.1%, and he between-run imprecisions were 1.5%-10.5% and 2.1%-4.1%, respectively. The carryover rates of the 12 items ranged from 0 to 2.63%. Results of liner verification test showed the correlation coefficients of PS, PC, AT-Ⅲ, and vWF:Ag were 0.982-0.988. Results of method comprisons showed the correlation coefficients of ACL TOP 700 and other coagulation analyzer were more than 0.975, and the recommended reference ranges of all the 12 items were appropriate for our laboratory. Conclusions ACL TOP 700 coagulation analyzer has a good performance in accuracy, imprecision, carryover rate, linear range, and method comparison. It is suitable for detection of clinical specimens. -
Key words:
- coagulation analyzer /
- thrombosis and homeostasis /
- performance
-
表 1 凝血因子、易栓症筛查三项和vWF : Ag准确度结果
项目 CGE1 CGE2 靶值 测定值 偏差(%) 靶值 测定值 偏差(%) 凝血因子 FⅡ 93.4 85.4 -8.57 35.5 33.2 -6.48 FⅤ 91.2 81.4 -10.75 33.6 31 -7.74 FⅦ 101.9 92.7 -9.03 34.5 31.7 -8.12 FⅧ 105.7 104.1 -1.51 163.2 166.4 1.96 FⅨ 115.7 117.4 1.47 45.7 47.9 4.81 FⅩ 97 89.4 -7.84 35.1 33.8 -3.7 FⅪ 88.3 93.8 6.23 36.4 38.2 4.95 FⅫ 101.8 97.2 -4.52 38.4 36 -6.25 易栓症筛 PS 93.4 101.1 8.24 32.1 28.5 -11.21 查三项 PC 97.5 85 -12.82 31.3 26 -16.93 AT-Ⅲ 86.6 91 5.08 30.4 31.1 2.3 vWF:Ag 158.2 167 5.56 240.7 214.8 -10.76 PS:蛋白S;PC:蛋白C;AT-Ⅲ:抗凝血酶-Ⅲ;vWF : Ag:血管性血友病因子抗原 表 2 凝血因子八项批内不精密度及批间不精密度
项目 批内不精密度 批间不精密度 水平1(x±s) CV(%) 水平2(x±s) CV(%) 水平1(x±s) CV(%) 水平2(x±s) CV(%) FⅡ 29.2±0.61 2.1 84.8±2.58 3.0 31.61±1.94 6.1 93.27±4.52 4.8 FⅤ 27.3±0.94 3.5 83.1±1.87 2.3 30.12±1.90 6.3 103.55±7.46 7.1 FⅦ 25.8±0.58 2.2 85.0±1.76 2.1 27.17±2.05 7.5 98.58±3.35 3.4 FⅧ 24.8±0.61 2.5 79.3±2.86 3.6 24.74±1.83 7.4 72.47±3.42 4.7 FⅨ 37.6±1.65 4.4 112.3±45.2 4.0 37.26±1.85 5.0 99.13±6.15 6.2 FⅩ 29.1±0.93 3.2 88.1±1.49 1.7 32.66±2.25 6.9 95.49±2.14 2.2 FⅪ 31.5±1.06 3.4 91.0±3.90 4.3 32.21±1.36 4.2 97.30±6.40 6.6 FⅫ 28.6±0.86 3.0 83.3±1.58 1.9 28.84±1.30 4.5 83.06±3.77 4.5 CV:变异系数 表 3 易栓症筛查三项及vWF : Ag批内不精密度及批间不精密度
项目 批内不精密度 批间不精密度 水平1(x±s) CV(%) 水平2(x±s) CV(%) 水平3(x±s) CV(%) 水平1(x±s) CV(%) 水平2(x±s) CV(%) 水平3(x±s) CV(%) PS 25.38±1.78 7.0 38.57±1.73 4.5 100.46±5.16 5.1 21.94±2.30 10.5 41.84±3.78 9.0 107.54±8.71 8.1 PC 12.10±0.32 2.6 24.50±1.18 4.8 94.50±0.97 1.0 13.36±0.45 3.4 32.45±1.01 3.1 103.79±1.57 1.5 AT-Ⅲ 14.10±0.88 6.2 23.90±0.88 3.7 110.40±1.07 1.0 15.04±0.98 6.5 31.09±1.62 5.2 111.17±2.03 1.8 vWF:Ag 28.71±0.90 3.1 60.17±1.63 2.7 106.47±2.34 2.2 30.33±1.13 3.6 59.95±1.24 2.1 103.63±4.23 4.1 PS、PC、AT-Ⅲ、vWF : Ag:同表 1;CV:同表 2 表 4 各脂肪酸与其他生化指标的相关系数
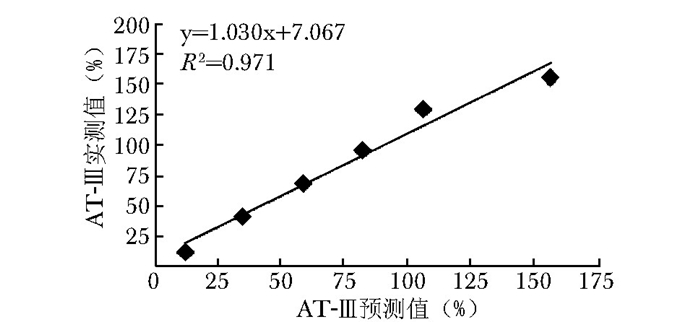
项目 r 偏差(%) 凝血因子 FⅡ 0.993 -8.5 FⅤ 0.983 15.5 FⅦ 0.998 3.7 FⅧ 0.975 3.9 FⅨ 0.993 -12.6 FⅩ 0.998 -11.7 FⅪ 0.989 7.7 FⅫ 0.990 7.4 易栓症筛查三项 PS 0.975 3.5 PC 0.993 3.3 AT-Ⅲ 0.983 15.2 vWF:Ag 0.975 9.3 PS、PC、AT-Ⅲ、vWF : Ag:同表 1 -
[1] National Committee for Clinical Laboratory Standards. Evaluation of precision performance of quantitative measurement methods; Approved guideline. EP5-A2[S]. Wayne, PA: NCCLS, 2004. [2] National Committee for Clinical Laboratory Standards. Method comparison and bias estimation using patient samples; Approved guideline. EP9-A2[S]. Wayne, PA: NCCLS, 2002. [3] 寿玮龄, 崔巍.恶性肿瘤出凝血异常机制[J].协和医学杂志, 2012, 4:482-486. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=xhyx201204026 [4] Appert-Flory A, Fischer F, Jambou D, et al. Evaluation and performance characteristics of the automated coagulation analyzer ACL TOP[J]. Thromb Res, 2007, 120:733-743. doi: 10.1016/j.thromres.2006.12.002 [5] 王海, 王成彬, 齐晓伟, 等.凝血功能分析的系统差异性对临床决策的影响探讨[J].中华临床医师杂志, 2012, 6:4371-4374. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zhlcyszz201215047 [6] Bai B, Christie DJ, Gorman RT, et al. Comparison of optical and mechanical clot detection for routine coagulation testing in a large volume clinical laboratory[J]. Blood Coagul Fibrinolysis, 2008, 19:569-576. doi: 10.1097/MBC.0b013e3283070872 [7] Dentali F, Sironi AP, Ageno W, et al. ABO blood group and vascular disease:an update[J]. Semin Thromb Hemost, 2014:49-59. http://www.ncbi.nlm.nih.gov/pubmed/24381150 -


 作者投稿
作者投稿 专家审稿
专家审稿 编辑办公
编辑办公 邮件订阅
邮件订阅 RSS
RSS


 下载:
下载: