-
摘要:
目的 体外检测卵巢癌患者血中CA125对细胞免疫功能的影响。 方法 收集2012年5月至2013年6月北京协和医院符合诊断标准的卵巢癌患者58例, 按CA125浓度分为3组; 同时收集良性妇科疾病患者20例及体检正常的健康者20名作为对照。应用细胞培养、酶联免疫吸附法、酶联免疫斑点(enzyme-linked immunospot, ELISPOT)等实验, 观察体外刺激健康人淋巴细胞产生γ-干扰素(interferon-γ, IFN-γ)的能力, 判断CA125对机体细胞免疫状态的影响。 结果 在卵巢癌患者中, 随CA125水平升高, 淋巴细胞绝对值及百分比值呈逐渐下降趋势。高浓度CA125卵巢癌患者的血清与植物血凝素共同刺激健康者混合淋巴细胞后, 培养上清液中IFN-γ的含量[(36.16±16.89)ng/ml]较阳性对照组和低浓度CA125组[分别为(45.68±18.01)和(46.22±19.30)ng/ml]显著降低(P < 0.05);ELISPOT结果显示高浓度和中浓度CA125(斑点数均值分别为108.2和371.6)对细胞免疫抑制功能与健康对照组和良性疾病组(斑点数均值分别为573.0和523.0)比较, 差异均有统计学意义(P < 0.01, P < 0.05);而去除血清中CA125后, 对细胞免疫抑制作用明显减弱。 结论 卵巢癌患者体内的CA125水平与淋巴细胞的数量、活性及其产生细胞因子的活性呈负相关关系。CA125除作为与卵巢癌相关的标志物外, 还可能是一种细胞免疫抑制因子。 Abstract:Objective To detect the effects of CA125 on cellular immune function of the ovarian cancer (OC) patients in vitro. Methods Totally 58 patients with confirmed OC in our hospital from May 2012 to June 2013 were divided into three groups based on the CA125 levels. Meanwhile, 20 healthy subjects(control group) and 20 patients with benign gynecological diseases (benign diseases group) were also included. The in vitro ability of healthy human lymphocytes in secreting interferon-γ (IFN-γ) under the stimulation of phytohemagglutinin(PHA) was observed by cell culture, enzyme-linked immunosorbent assay (ELISA) and enzyme-linked immunospot (ELISPOT). Results In OC patients, the count and percentage of lymphocytes decreased gradually with elevated levels of CA125. The concentration of IFN-γ in the supernatant of cultured lymphocytes from healthy individuals decreased significantly under the stimulation of PHA and high concentration of CA125[(36.16±16.89)ng/ml]compared with that of PHA alone [(45.68±18.01)ng/ml] or PHA and low concentration of CA125 [(46.22±19.30)ng/ml](both P < 0.05). Compared with the control group(spots number 573.0) and the benign diseases group(spots number 523.0), the high(spots number 108.2) and intermediate(spots number 371.6) concentration of CA125 had significant inhibitory effect on the cellular immune system, as detected by ELISPOT (P < 0.01, P < 0.05, respectively). After removal of CA125 from the serum by antibody, the inhibitory effect was weakened significantly. Conclusions The concentration of CA125 in OC patient is negatively correlated with the number and activity of lymphocytes, in particular their abilities in releasing cytokines. CA125 is not only a biomarker of OC but also may serve as an immunosuppressive factor. -
Key words:
- CA125 /
- ovarian cancer /
- lymphocyte /
- cytokine /
- interferon-γ
-
表 1 不同浓度CA125卵巢癌患者外周血淋巴细胞数量和比率(x±s)
组别 CA125
(U/ml)淋巴细胞
(×109/L)淋巴细胞
比率(%)对照组 健康对照组(n=20) 11.2±5.0 1.43±0.28 31.1±6.2 良性疾病组(n=20) 25.8±26.8 1.75±0.71 32.2±11.0 疾病组 高浓度组(n=26) 5032±1850 1.27±0.46* 21.1±13.5* 中浓度组(n=19) 1559±566 1.39±0.57 25.4±10.6 低浓度组(n=13) 526±196 1.53±0.60 26.2±12.9 与健康对照组和良性疾病组比较,*P<0.05 表 2 不同浓度CA125对培养T淋巴细胞免疫抑制作用比较
组别 CA125
(U/ml)ELISPOT
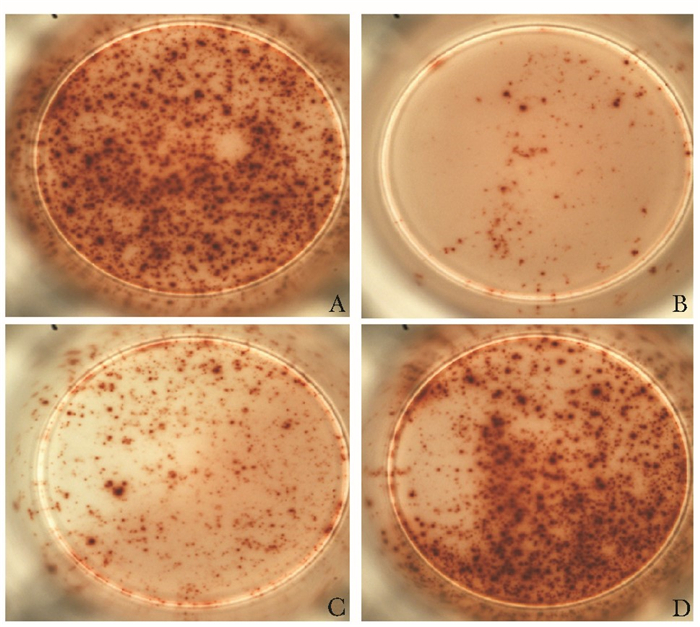
斑点数均值抑制率
(%)对照组 健康对照组(n=5) 15 573.0 0 良性疾病组(n=5) 30 523.0 8.7 疾病组 高浓度组(n=5) 3000 108.2** 81.1** 中浓度组(n=5) 1000 371.6* 35.1* 低浓度组(n=5) 300 528.8 7.8 ELISPOT:酶联免疫斑点实验;与健康对照组和良性疾病组比较,*P<0.05,* *P<0.01 表 3 部分血清去除CA125后的免疫相关检测结果
病例 CA125
(U/ml)抑制率(%) γ-干扰素(ng/ml) 原浓度 去除后 原浓度 去除后 原浓度 去除后 1 3106 309.9 86.2 84.8 37.3 44.6 2 3185 320.8 78.1 40.0 8.3 10.2 3 1727 389.3 87.4 84.9 54.0 55.5 -
[1] Kobayashi E, Ueda Y, Matsuzaki S, et al. Biomarkers for screening, diagnosis, and monitoring of ovarian cancer[J]. Cancer Epidemiol Biomarkers Prev, 2012, 21:1902-1912. doi: 10.1158/1055-9965.EPI-12-0646 [2] Rooth C. Ovarian cancer:risk factors, treatment and management[J]. Br J Nurs, 2013, 22:S23-S30. doi: 10.12968/bjon.2013.22.Sup18.S23 [3] Yin BW, Lloyd KO. Molecular cloning of the CA125 ovarian cancer antigen:identification as a new mucin, MUC16[J]. J Biol Chem, 2001, 276:27371-27375. doi: 10.1074/jbc.M103554200 [4] Gubbels JA, Felder M, Horibata S, et al. MUC16 provides immune protection by inhibiting synapse formation between NK and ovarian tumor cells[J]. Mol Cancer, 2010, 9:11. doi: 10.1186/1476-4598-9-11 [5] Belisle JA, Gubbels JA, Raphael CA, et al. Peritoneal natural killer cells from epithelial ovarian cancer patients show an altered phenotype and bind to the tumour marker MUC16(CA125)[J]. Immunology, 2007, 122:418-429. doi: 10.1111/j.1365-2567.2007.02660.x [6] Patankar MS, Jing Y, Morrison JC, et al. Potent suppression of natural killer cell response mediated by the ovarian tumor marker CA125[J]. Gynecol Oncol, 2005, 99:704-713. doi: 10.1016/j.ygyno.2005.07.030 [7] Lanier LL. Natural killer cell receptor signaling[J]. Curr Opin Immunol, 2003, 15:308-314. doi: 10.1016/S0952-7915(03)00039-6 [8] Tyler C, Kapur A, Felder M, et al. The mucin MUC16(CA125) binds to NK cells and monocytes from peripheral blood of women with healthy pregnancy and preeclampsia[J]. Am J Reprod Immunol, 2012, 68:28-37. doi: 10.1111/j.1600-0897.2012.01113.x [9] Radka S, Weston BS, Kieran W, et al. Exploring the glycosylation of serum CA125[J]. Int J Mol Sci, 2013, 14:15636-15654. doi: 10.3390/ijms140815636 [10] 朱兰, 郎景和.肿瘤标记物在妇科领域中的应用[J].中国实验诊断学, 2012, 6:301-302. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zgsyzdx200205029 [11] Gipson IK, Blalock T, Tisdale A, et al. MUC16 is lost from the uterodome (pinopode) surface of the receptive human endometrium:in vitro evidence that MUC16 is a barrier to trophoblast adherence[J]. Biol Reprod, 2007, 78:134-142. [12] Pfisterer J, Mahner S. Towards individualised treatment in ovarian cancer[J]. Lancet Oncol, 2013, 14:101-102. doi: 10.1016/S1470-2045(13)70006-8 -


 作者投稿
作者投稿 专家审稿
专家审稿 编辑办公
编辑办公 邮件订阅
邮件订阅 RSS
RSS


 下载:
下载: