-
摘要:
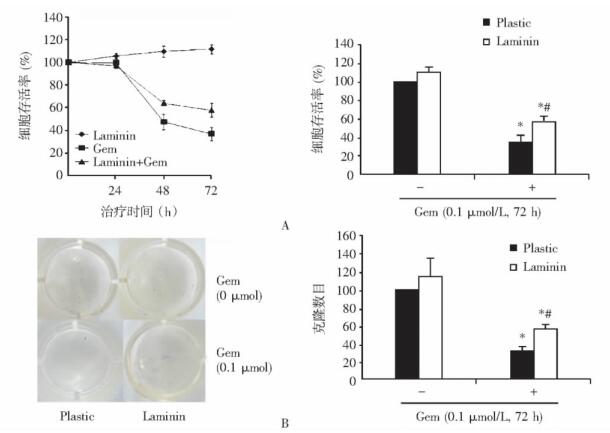
目的 探讨层黏连蛋白(laminin, LN)对胰腺癌细胞内在性耐药的影响, 并探讨其作用机制。 方法 选择胰腺癌细胞系AsPC-1, 检测LN对盐酸吉西他滨(健择, gemcitabine, Gem)的细胞毒性及对Gem诱导细胞凋亡能力的影响。通过Western blot等方法检测LN对下游信号通路的影响; 在AsPC-1中, 过表达黏着斑激酶相关非激酶(focal adhesion kinase-related non-kinase, FRNK)或应用PF-573, 228抑制黏着斑激酶(focal adhesion kinase, FAK)磷酸化与活性, 检测FAK及其下游信号的抑制对LN作用的影响。 结果 在胰腺癌细胞系AsPC-1中, LN处理使Gem的细胞毒性及Gem诱导细胞凋亡的能力显著降低。Gem作用72 h后, LN处理组细胞存活率为57.71%±6.08%, 形成的克隆数目为55.33±5.01;而在无LN包被组, 细胞存活率与形成的克隆数目分别为36.65%±4.14%及31.43±4.62(P均 < 0.05);LN处理组Annexin V标记阳性细胞百分数(41.00%±5.46%)较无LN处理组(55.70%±3.44%)显著下降(P < 0.05)。LN能够时间依赖性上调AsPC-1细胞的FAKTyr397位点及Akt磷酸化水平, LN还可以显著增加AsPC-1细胞的survivin蛋白表达水平及BAD Ser-136位点磷酸化水平。过表达FRNK或应用PF-573, 228能够显著抑制FAK磷酸化水平及其下游通路的活化, 并能够对抗LN对胰腺癌内在性耐药的效应。应用PF-228可以使LN处理后Gem诱导的AsPC-1细胞凋亡率从26.77%±0.49%升高至38.53%±2.83%(P < 0.05)。 结论 细胞外基质蛋白LN能够诱导胰腺癌细胞系对Gem产生内在性耐药。LN影响胰腺癌细胞内在性耐药的具体机制可能与FAK磷酸化及其下游PI3K-Akt通路活化、Bad Ser-136位点磷酸化与survivin表达水平改变有关, FAK靶向治疗与Gem联合在胰腺癌治疗中具有重要的潜在应用价值。 Abstract:Objective To determine the effect of laminin (LN) on the intrinsic chemoresistance of pancreatic cancer and whether such effect is mediated by LN-induced focal adhesion kinase (FAK) phosphorylation and subsequent downstream signal pathway. Methods The effect of LN on gemcitabine (Gem) -induced cytotoxicity and apoptosis in pancreatic cancer cell line AsPC-1 cells were determined by MTT assay, clonogenic assay and apoptosis analysis. The effect of LN on the expression and phosphorylation of FAK, Akt, and ERK1/2 were detected by Western blotting. The changes in the effects of LN in AsPC-1 cells were explored by FAK phosphorylation inhibition through focal adhesion kinase-related non-kinase (FRNK) overexpression or specific FAK phosphorylation inhibitor PF-573, 228. Results LN decreased Gem-induced cytotoxicity and apoptosis in AsPC-1 cells. After Gem treatment for 72 h, the viability was 57.71%±6.08% and the colony number was 55.33±5.01 on LN, while the viability and the colony number on plastic were 36.65%±4.14% and 31.43±4.62, respectively (both P < 0.05). The Annexin V positivity of AsPC-1 cells on LN (41.00%±5.46%) was significantly lower than that on plastic (55.70%±3.44%) (P < 0.05). Moreover, LN induced FAK and Akt phosphorylation in a time-dependent manner and increased the levels of survivin and pBad (pS136). Specific inhibition of LN-induced FAK phosphorylation by either FRNK overexpression or PF-573, 228 suppressed the effect of LN on AsPC-1 cells. PF-573, 228 increased Gem-induced apoptosis in AsPC-1 cells from 26.77%±0.49% to 38.53%±2.83% on LN (P < 0.05). Conclusions LN contributes to the increased intrinsic chemoresistance of pancreatic cancer cell to Gem, which may be achieved through the regulation of FAK, Akt, and Bad phosphorylation and survivin expression. Targeted FAK inhibitors may be a promising way to enhance chemosensitivity in pancreatic cancer. -
Key words:
- laminin /
- pancreatic cancer /
- chemoresistance /
- focal adhesion kinase
-
图 2 层黏蛋白对Gem诱导AsPC-1细胞凋亡的效应
A. Hoechst 33342染色; B.流式细胞术检测Annexin V标记; C. Western blot检测caspase-3蛋白剪切
Plastic, Laminin, Gem:同图 1; 与无Gem处理各组比较, * P < 0.05;与有Gem处理Plastic组比较, #P < 0.05图 3 PF-228对层黏蛋白诱导的凋亡相关蛋白表达与磷酸化改变的影响
Plastic, Laminin:同图 1
图 4 层黏蛋白对FAK及其下游激酶Akt与ERK1/2表达与磷酸化的影响
Laminin:同图 1
图 5 FRNK过表达及PF-228对层黏连蛋白诱导FAK及Akt磷酸化的影响
Plastic, Laminin:同图 1; Nontransfected:空白转染; Vector:对照质粒转染; FRNK: FRNK表达质粒转染
图 6 PF-228对层黏连蛋白诱导的AsPC-1细胞对Gem内在性耐药的影响
A. Hoechst 33342染色; B.流式细胞术检测Annexin V标记; C. Western blot检测caspase-3蛋白剪切
Plastic, Laminin:同图 1; 与无Gem处理各组比较, * P < 0. 05;与有Gem处理而无PF-228作用Laminin组比较, #P < 0. 05 -
[1] Li D, Xie K, Wolff R, et al. Pancreatic cancer[J]. Lancet, 2004, 363:1049-1057. doi: 10.1016/S0140-6736(04)15841-8 [2] von Wichert G, Seufferlein T, Adler G. Palliative treatment of pancreatic cancer[J]. J Dig Dis, 2008, 9:1-7. doi: 10.1111/j.1443-9573.2007.00314.x [3] Meads MB, Gatenby RA, Dalton WS. Environment-mediated drug resistance:a major contributor to minimal residual disease[J]. Nat Rev Cancer, 2009, 9:665-674. doi: 10.1038/nrc2714 [4] Meads MB, Hazlehurst LA, Dalton WS. The bone marrow microenvironment as a tumor sanctuary and contributor to drug resistance[J]. Clin Cancer Res, 2008, 14:2519-2526. doi: 10.1158/1078-0432.CCR-07-2223 [5] Hodkinson PS, Elliott T, Wong WS, et al. ECM overrides DNA damage-induced cell cycle arrest and apoptosis in small-cell lung cancer cells through beta1 integrin-dependent activation of PI3-kinase[J]. Cell Death Differ, 2006, 13:1776-1788. doi: 10.1038/sj.cdd.4401849 [6] Cordes N, Blaese MA, Plasswilm L, et al. Fibronectin and laminin increase resistance to ionizing radiation and the cytotoxic drug Ukrain in human tumour and normal cells in vitro[J]. Int J Radiat Biol, 2003, 79:709-720. doi: 10.1080/09553000310001610240 [7] Miyamoto H, Murakami T, Tsuchida K, et al. Tumor-stroma interaction of human pancreatic cancer:acquired resistance to anticancer drugs and proliferation regulation is dependent on extracellular matrix proteins[J]. Pancreas, 2004, 28:38-44. doi: 10.1097/00006676-200401000-00006 [8] Sawai H, Funahashi H, Matsuo Y, et al. Expression and prognostic roles of integrins and interleukin-1 receptor type Ⅰ in patients with ductal adenocarcinoma of the pancreas[J]. Dig Dis Sci, 2003, 48:1241-1250. doi: 10.1023/A:1024276821731 [9] Nolan K, Lacoste J, Parsons JT. Regulated expression of focal adhesion kinase-related nonkinase, the autonomously expressed C-terminal domain of focal adhesion kinase[J]. Mol Cell Biol, 2003, 19:6120-6129. http://europepmc.org/articles/PMC84535 [10] Slack-Davis J, Tilghman R, Roberts WG. Focal adhesion kinase:targeting adhesion signaling pathways for therapeutic intervention[J]. Clin Cancer Res, 2008, 14:627-632. doi: 10.1158/1078-0432.CCR-07-2220 [11] Wang Z, Li Y, Ahmad A, et al. Pancreatic cancer:understanding and overcoming chemoresistance[J]. Nat Rev GastroenterolHepatol, 2010, 8:27-33. http://www.ncbi.nlm.nih.gov/pubmed/21102532/ [12] Kouniavsky G, Khaikin M, Zvibel I, et al. Stromal extracellular matrix reduces chemotherapy-induced apoptosis in colon cancer cell lines[J]. Clin Exp Metastasis, 2002, 19:55-60, 33. doi: 10.1023/A:1013880326925 [13] Pan C, Shen Z, Wu T, et al. Cell adhesion to fibronectin induces mitomycin C resistance in bladder cancer cells[J]. BJU Int, 2009, 104:1774-1779. doi: 10.1111/j.1464-410X.2009.08639.x [14] Dangi-Garimella S, Krantz, SB, Barron MR, et al. Threedimensional collagen I promotes gemcitabine resistance in pancreatic cancer through MT1-MMP-mediated expression of HMGA2[J]. Cancer Res, 2011, 71:1019-1028. doi: 10.1158/0008-5472.CAN-10-1855 [15] Pandol S, Edderkaoui M, Gukovsky I, et al. Desmoplasia of pancreatic ductal adenocarcinoma[J]. Clin Gastroenterol Hepatol, 2009, 7:S44-S47. doi: 10.1016/j.cgh.2009.07.039 [16] Armstrong T, Packham G, Murphy LB, et al. type Ⅰ collagen promotes the malignant phenotype of pancreatic ductal adenocarcinoma[J]. Clin Cancer Res, 2004, 10:7427-7437. doi: 10.1158/1078-0432.CCR-03-0825 [17] Bouchard V, Harnois C, Demers MJ, et al. B1 integrin/Fak/Src signaling in intestinal epithelial crypt cell survival:integration of complex regulatory mechanisms[J]. Apoptosis, 2008, 13:531-542. doi: 10.1007/s10495-008-0192-y [18] Fong YC, Liu SC, Huang CY, et al. Osteopontin increases lung cancer cells migration via activation of the alphavbeta3 integrin/FAK/Akt and NF-kappaB-dependent pathway[J]. Lung Cancer, 2008, 64:263-270. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=9274083da3f4e6527150b83df12d4381 [19] Parsons JT. Focal adhesion kinase:the first ten years[J]. J Cell Sci, 2003, 116:1409-1416. doi: 10.1242/jcs.00373 [20] Bouchard V, Harnois C, Demers MJ, et al. B1 integrin/Fak/Src signaling in intestinal epithelial crypt cell survival:integration of complex regulatory mechanisms[J]. Apoptosis, 2008, 13:531-542. doi: 10.1007/s10495-008-0192-y [21] Rückert F, Samm N, Lehner AK, et al. Simultaneous gene silencing of Bcl-2, XIAP and Survivin re-sensitizes pancreatic cancer cells towards apoptosis[J]. BMC Cancer, 2010, 10:379-388. doi: 10.1186/1471-2407-10-379 [22] Werner K, Rückert F, Saeger HD, et al. Recent patents concerning targeted therapy of apoptosis resistance in pancreatic cancer[J]. Recent Pat DNA Gene Seq, 2011, 5:28-34. doi: 10.2174/187221511794839237 [23] Hayakawa J, Ohmichi M, Kurachi H, et al. Inhibition of BAD phosphorylation either at serine 112 via extracellular signal-regulated protein kinase cascade or at serine 136 via Akt cascade sensitizes human ovarian cancer cells to cisplatin[J]. Cancer Res, 2000, 60:5988-5994. http://www.ncbi.nlm.nih.gov/pubmed/11085518 [24] Jiang C, Tan T, Yi XP, et al. Lentivirus-mediated shRNA targeting XIAP and survivin inhibit SW1990 pancreatic cancer cell proliferation in vitro and in vivo[J]. Mol Med Report, 2011, 4:667-674. http://www.ncbi.nlm.nih.gov/pubmed/21479361 -


 作者投稿
作者投稿 专家审稿
专家审稿 编辑办公
编辑办公 邮件订阅
邮件订阅 RSS
RSS


 下载:
下载: